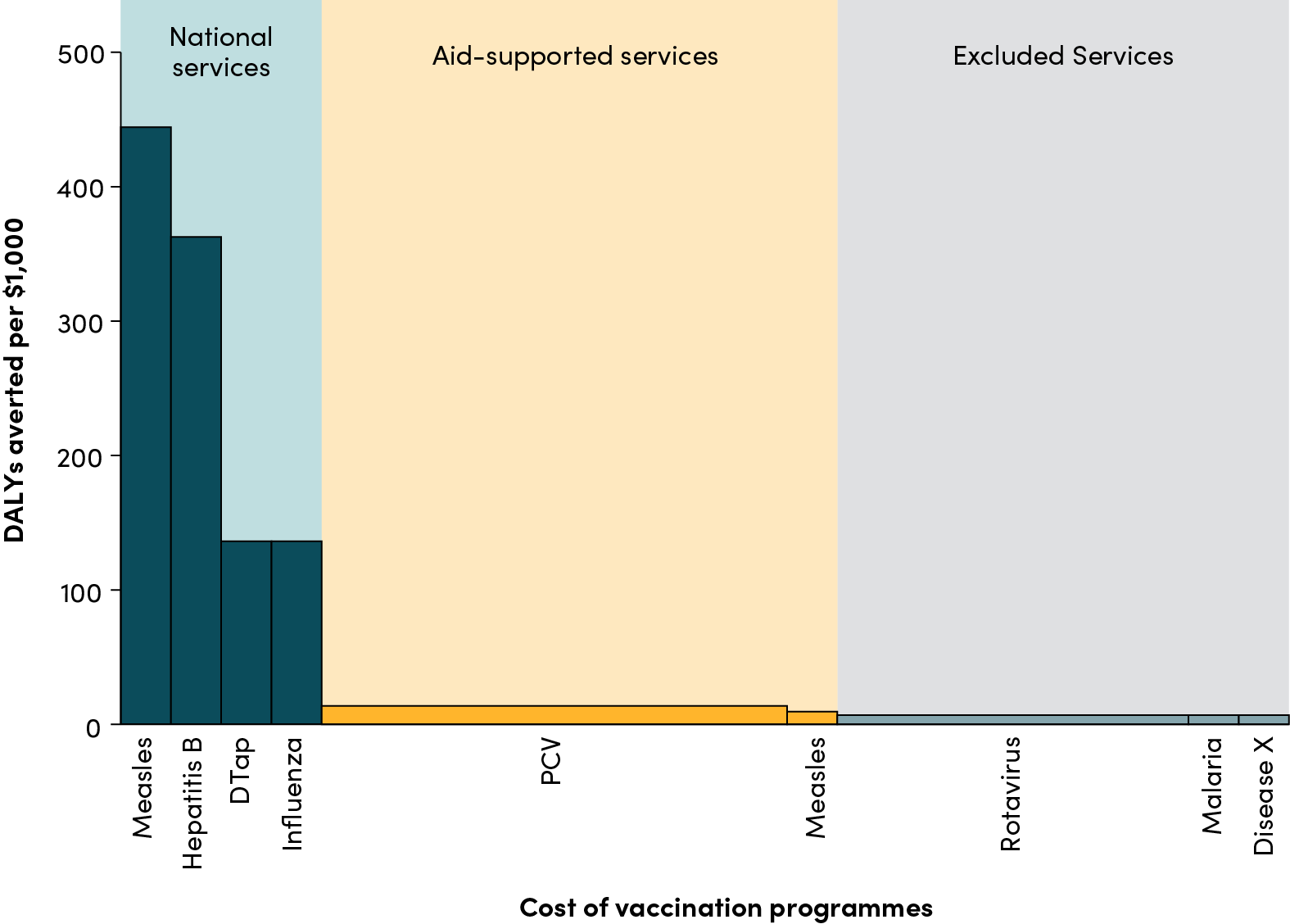
Recommended

Effective and efficient procurement of health products—medicines, diagnostics, and devices—is a critical function of all strong health systems. The pandemic has exacerbated long-standing challenges—as highlighted by a recent CGD Working Group—in the purchasing of both COVID-related emergency supplies and other essential health products. Procurement challenges in the context of COVID-19 range from disruptions to sourcing active pharmaceutical ingredients, export restrictions, and transport interruptions to quality assurance issues, reduced household income to cover out of pocket spending, and domestic financing pressures amidst uncertainty in the future of health aid.
To harness COVID-19 as a catalyst for better procurement practices, the World Bank’s Health Financing Global Solutions Group, the Global Financing Facility, and the Joint Learning Network’s Efficiency Collaborative hosted an online event in early July. The webinar, chaired by one of us (Chalkidou), brought together experts from the Philippines and Thailand to showcase ongoing improvements to national procurement systems, alongside presentations of CGD research from two of us (Yadav and Madan Keller) on broader opportunities to deliver access to affordable, high-quality health products. Here are three takeaways.
1. Collaborate across countries to pool procurement
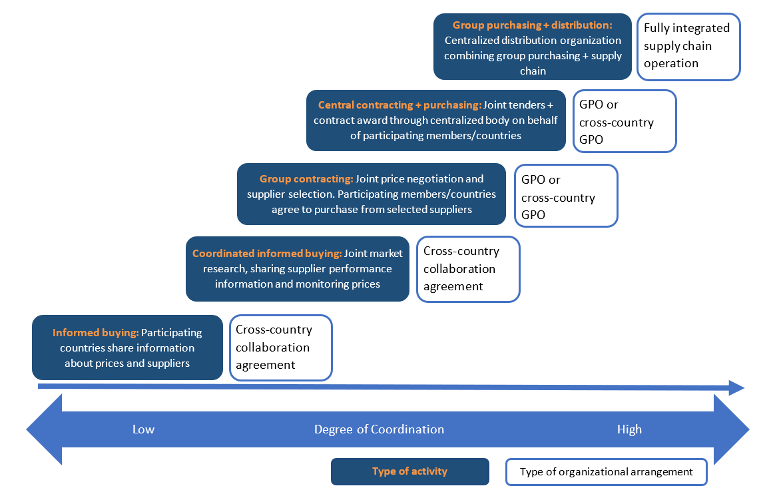
Amidst uncoordinated scrambles for emergency supplies, hoarding, and even bidding wars, the pandemic has put the importance of achieving economies of scale in the limelight. Fragmented demand across purchasing entities can lead to higher transaction costs to serve these markets, which can often be passed on to purchasers and eventually to consumers. Pooled procurement or other forms of cooperative purchasing across countries offers one way to aggregate demand, increase purchaser bargaining power, and better organize procurement-related functions like horizon scanning and information-sharing about the supplier landscape (see Figure 1).
The Africa CDC’s newly launched Africa Medical Supplies Platform (AMSP)—an online marketplace that pools orders for COVID-19 diagnostics and medical equipment—is a promising development with ample potential. But long-standing challenges stemming from myriad political, legal, and regulatory barriers (e.g., national industrial policies that favor domestic suppliers or the desire to exercise country preferences in product choice) will need to be tackled for such initiatives to have lasting benefits.
Whether this platform goes beyond COVID-19 to successfully facilitate purchasing of other essential medicines at scale and effectively incorporates related functions such as health technology assessment remains to be seen. As the AMSP continues to evolve, different pooling mechanisms at the regional-level alongside donor-supported mechanisms at the global level, each with diverse organizational arrangements, could offer lessons about what has and has not worked. These collaborative purchasing efforts will also be increasingly critical as middle-income countries navigate health financing transitions, as emphasized by CGD’s Working Group. The AMSP and other COVID-19 procurement initiatives like the ACT Accelerator also elevate the importance of coordination and information-sharing from the get-go to identify gaps and avoid overlap.
Figure 1. Different pooling activities and types of organizational arrangements
Source: Nemzoff, Chalkidou, and Over 2019, adapted from Espín et al. 2016.
2. Embed resilience in procurement contracting
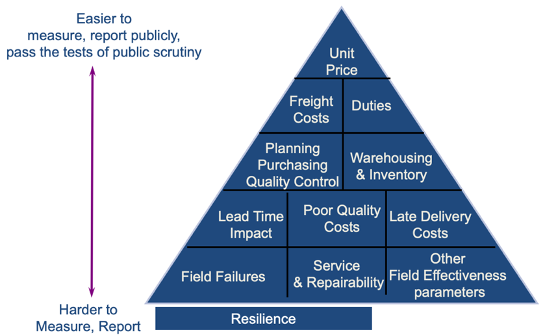
COVID-19 has illustrated that buying from the lowest-cost supplier may come at the expense of other procurement outcomes like quality, supply security, speed, or resilience. Some suppliers may be able to guarantee or demonstrate a degree of resilience based on past performance—such as the ability to meet a certain portion of a demand surge within a guaranteed lead time. But not all buyers may be willing to pay higher prices to prepare for and respond to crises, reflecting a classic public good problem as colleagues have argued. Nevertheless, this specific metric is just one potential example; other relevant dimensions of resilience include resilience to production disruption and resilience to sourcing scarce raw material. In reality, purchasers tend to lack granular, applicable metrics and sufficient data to assess and maximize relevant resilience attributes as part of supplier selection (see Figure 2). More and better empirical evidence on how to measure resilience before stress is applied to the system is needed.
Further, while open contracting can expedite efficient procurement, these approaches need to be balanced with assuring that procurement regulations and scrutiny do not become obstacles to assessing and incorporating resilience into contracting decisions. Procurement agents should have the flexibility to comprehensively consider various dimensions of resilience and responsiveness as part of bid evaluation decisions in order to sustain and expand health product access in times of crisis (and “normal” times as well).
Another tactical area of procurement altered by COVID-19 is split awards. At the national level, these awards may need to more explicitly incorporate ways to split overall procurement volume between multiple suppliers. And while many are interested in “shortening” supply chains, geographic diversification can help to address potential supply chain risks and disruptions down the line. Recommendations on the ideal number of suppliers for each product category and the best delivery frequency to buffer stocks are still evolving, but some experts have recommended buying smaller quantities more frequently where possible, with weekly deliveries as opposed to annual ones. Better resilience heuristics, flexible governance arrangements, and nuanced financing regulations can enable more resilient global health procurement going forward.
Figure 2. Supplier Selection Attributes
Source: Prashant Yadav
3. Consider the optimal locus of specific procurement functions
Procurement involves a gamut of functions, extending from market intelligence, technology horizon scanning and product selection to price negotiation, ordering, contract management, and performance monitoring. Determining the optimal locus of specific procurement functions requires careful consideration of tradeoffs between local agility and information versus economies of scale and scope.
Large national purchasers may be better suited to certain procurement-related functions, such as conducting market intelligence and price negotiation. For others, such as placing orders under negotiated contract terms, subnational actors might be better positioned. Without local information and knowledge, purchasing decisions are at risk of being removed from the relevant health system context, as has occurred with the so-called medical equipment graveyard.
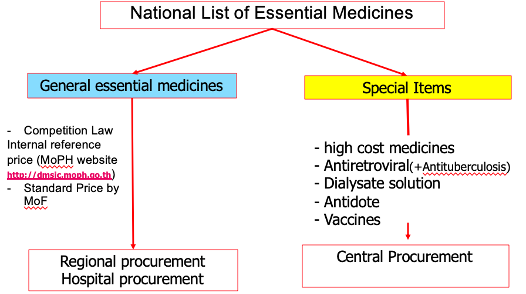
During the event, Somruethai Supungul of Thailand’s National Health Security Office outlined how regional and hospital-level procurement are used for broad essential medicines. However, central procurement processes are utilized for COVID-19 products and other special items. This approach helps to strike a balance between ensuring good value-for-money while also empowering facilities with the flexibility to purchase the products they know they need for their populations (see Figure 3). Jude Daval-Santos of the Philippines also explained that the country has centralized the procurement of COVID-19 laboratory supplies to remove the burden of dealing with negotiations from laboratories.
Although national and sub-national procurement can go hand in hand, with national agencies negotiating prices and accrediting suppliers alongside provincial or local actors placing purchase orders, this strategy faces political economy, process-related, and PFM issues that require more evidence to address. Speakers also emphasized that robust health information and logistics management systems are necessary to drive effective decision-making at different points all along the supply chain. Supungul highlighted the vital role of health information systems in delaying stockout impacts in Thailand, and Daval-Santos described the development of a digital dashboard to synthesize information on commodities for laboratories. Relatedly, COVID-19 has underscored the advantages of innovative digital marketplaces for rapid visibility into fluctuating stock levels and prices, such as such as Maisha Meds, MedSource, mPharma, and Pharmarack.
Figure 3. System to Increase the Accessibility of Special Medicines in Thailand
Source: National Health Security Office of Thailand
Investing in Procurement Yields Lasting Returns
COVID-19 presents a window of opportunity for transformational change in procurement, including by achieving efficiencies that can be (re)allocated to the health sector and advance universal health coverage. Strengthening procurement systems is a worthwhile investment of scarce resources for country governments and development partners alike, which is especially important given the dire economic consequences of the pandemic and shrinking budgetary space for health. We recognize that analyzing how to better buy health products is a pretty wonky pursuit. But leveraging the current moment to mobilize lasting procurement reforms will go a long way in assuring that everyone around the world has access to high-quality, affordable medicines. That sounds pretty compelling to us.
Disclaimer
CGD blog posts reflect the views of the authors, drawing on prior research and experience in their areas of expertise. CGD is a nonpartisan, independent organization and does not take institutional positions.
Image credit for social media/web: ABBAS Farzami / Rumi Consultancy / World Bank