Recommended
Widely cited figures say that hundreds of millions of children are suffering from lead poisoning…
You’ve seen the headline; indeed, you’ve probably seen it from us. According to widely cited estimates, about one in three children around the world are lead-poisoned, or about 800 million total. This means that they have blood-lead levels exceeding 5 micrograms per deciliter, a common reference level for lead exposure that requires health intervention to address. Almost all of these children live in low- and middle-income countries.
Lead poisoning is very bad, in many different ways and for many different reasons. It mounts a multi-pronged and permanent attack on children’s health, especially the brain and development during their vulnerable and formative early years, with life-long effects: cognitive deficits, lower educational attainment, and behavioral disorders.
The good news is that we think this is a problem which can be solved. It should be possible to eliminate lead poisoning among children—in fact, most high-income countries have almost already done so. But to do this, we need to get serious about measuring the problem.
…But we don’t know nearly enough about how many children are being poisoned, where, how and why
If you’re anything like us, a headline that big will send you in search of the small print. And what you’ll find is… not much. A recent systematic review found zero recent data on blood-lead levels for almost two-thirds of the world’s low- and middle-income countries (Ericson et al 2021).
So what do you use if you’re in a country that doesn’t have any recent direct data? The answer is imputation—often relying on some heroic assumptions—to produce estimates for countries and regions without local data, typically based on the characteristics of their population and other risk factors. Even a rough estimate can be better than nothing if the goal is to get policymakers’ attention and start a conversation. But at the risk of stating the obvious, guesstimates are not real data—even if they’re the best we can do for now given the patchy data landscape.
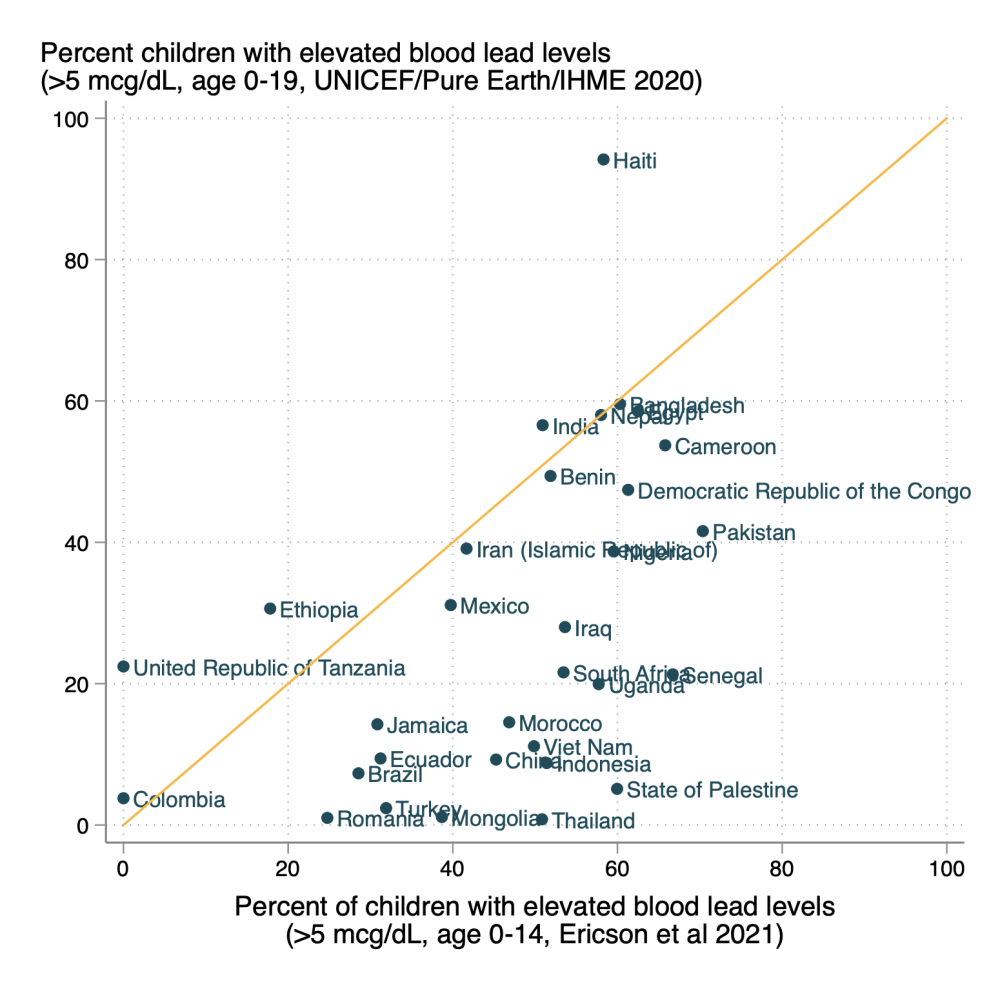
Even where data exist, samples are often unrepresentative. This leads to stark disagreements about national averages, as illustrated by two widely-cited sources for lead exposure in the developing world. Country-level estimates from the Institute for Health Metrics and Evaluation (IHME) at the University of Washington—which formed the basis of that “one in three kids”—differ substantially from estimates in the same countries reported by Erikson et al. (2021), as shown in Figure 1. These discrepancies show how much uncertainty we face due to small, unrepresentative samples.
Figure 1. Leading cross-national data sources disagree on levels of blood lead poisoning
Furthermore, some attempts to actually find nationally representative samples have found far higher prevalence of lead poisoning than suggested by the IHME estimates. For example, a nationally representative household survey conducted by the Government of Georgia with UNICEF support found a 41 percent prevalence of elevated blood lead levels (≥ 5 μg/dl) in children aged 2-7 years old, about 35 percentage points higher than the IHME point estimate (6 percent). This alone is a problem. We cannot say, with any real confidence, how large the global lead poisoning crisis really is, or where it ranks among health and education challenges, either at the global or local levels. And without real local data, national and regional policymakers may be understandably skeptical about whether to treat lead poisoning as an urgent, top-of-agenda priority. We should perhaps be particularly concerned if, like in Georgia, the extrapolated estimates are under-estimates, and too little action is taken because policymakers don’t realize how bad the problem is.
But the problem extends deeper. Without representative sub-national data, we also have no way of understanding which communities have been impacted or the sources of exposure or how we can effectively intervene. The data vacuum also creates a research vacuum—our literature review found 67 total studies linking lead exposure to socioeconomic and learning outcomes, but just two of those came from low- and middle-income country settings. Imputed data is also unsatisfactory when it comes to measuring progress on eliminating lead exposure. It’s unclear whether the models being used to do imputation can accurately measure improvements over time, and allow policymakers to assess their efforts.
In short, if we want to accurately measure lead poisoning in the majority of the world’s children, understand where it’s coming from (paint, battery recycling, spices, cosmetics, traditional medicine), or track progress in fixing it, we need real data not imputations.
Nationally representative blood-lead testing is part of routine surveillance in most high-income countries
Regular measurement of blood-lead levels, including via population-based surveys, is standard practice and a routine function of public health agencies. Public Health England, for example, publishes an annual surveillance report on lead exposure in children,that identifies cases of children resident in England with elevated blood lead concentrations, initiates timely public health action for individual cases, and informs public health interventions to prevent further exposures. In the US, state health departments conduct regular blood-lead surveillance, as well as surveillance in workplaces, water, and food. In addition, the US federal government (CDC) collects nationally representative data on blood-lead levels through its annual, rolling National Health and Nutrition Examination Survey (NHANES).
A 1977 EU Council Directive requires member states to do routine screening on lead exposure outside of the workplace (there’s a separate directive for workplace exposure). In Germany, for example, the Environmental Specimen Bank has been monitoring lead exposure in air and blood since 1981.
What all this means is that almost all high-income countries have a handle on the lead poisoning problems within their borders: they know the extent of the problem, its geographic and sociodemographic dimensions, and the underlying sources of lead exposure that affect their children. This robust surveillance has helped rich countries to address and dramatically reduce the effects of lead exposure in their populations over the past 50 years (Figure 2). In the US average blood-lead levels in children have fallen from 15 μg/dl in the 1970s to close to zero in 2016.
Figure 2. Lead concentration in the the blood of children in the US over time
Source: Centers for Disease Control and Prevention; National Center for Health Statistics, via Our World in Data
The experience of high-income countries demonstrates that it is possible to eliminate lead poisoning in children—but only if you know how and why it’s happening, and commit to doing better for kids’ welfare. We may have almost eliminated lead exposure in high incomes, but as communities in Flint, Michigan know only too well, its legacy lasts.
UNICEF and USAID have both piloted blood-lead testing within existing household surveys, but R&D is required to make it more practical and affordable
The good news is that there is a way to increase visibility into the global lead poisoning crisis: by incorporating a blood lead testing module into the existing, routine household surveys—specifically the Demographic and Health Surveys (DHS) sponsored by USAID and the Multiple Indicators Cluster Survey (MICS) supported by UNICEF, that form the backbone of research and evidence-based policy in many developing countries. These surveys are regularly conducted across many countries in Africa, Asia, and Latin America (Figure 3). In the last five years alone, nationally representative MICS or DHS surveys have been administered in 87 countries, 55 of which have included blood testing with finger pricks.
Figure 3. DHS and MICS surveys fielded since 2017, with and without blood testing
Blood testing is used in many of these surveys already. DHS often collects biomarker data using rapid, finger-stick tests for anemia, malaria, HIV, and high blood pressure. While MICS typically does not include biomarkers, it does when governments request it. Ghana’s 2011 survey, for example, included an enhanced malaria module at the request of the government following a recent malaria outbreak.
And, there’s precedent for blood lead testing within these surveys. A blood lead testing module was part of the MICS survey in Georgia in 2018, and DHS surveys in India in 1998 and Uzbekistan in 2002. In India and Uzbekistan, blood samples were obtained using a portable lead testing machine with capillary finger pricks. In Georgia, samples of venous blood were collected by phlebotomists and sent to the Italian National Institute of Health for analysis.
The choice between finger prick testing on portable machines and venous samples for laboratory analysis is a challenging one. Finger prick tests are far easier to administer and indeed many of the DHS surveys already have measurers or phlebotomists trained in sampling this way. But concerns have been raised about the accuracy of portable finger stick testing for lead, including the type used in the DHS surveys in India and Uzbekistan. While this appears to have been resolved recently, it’s recommended that any samples with high blood lead levels are sent to laboratories for validation. And environmental contamination is a high risk for finger prick testing and could limit its widespread use.
On the other hand, while venous samples with laboratory analysis are the gold standard for blood lead level testing, collecting venous samples from children requires a high degree of phlebotomy skill and appropriate laboratory capacity. Dried blood sampling from finger pricks-which are then sent to laboratories for analysis-is a method of blood lead level testing that seems to hold potential and should be tested more widely, although environmental contamination remains a risk.
What’s clear is that investment is needed in rapid diagnostic innovation and better laboratory capacity in developing countries, as well as consensus on what kind of testing is most suitable in which circumstances.
Real data opens up real opportunities
Nationally representative household surveys have been the basis for global and domestic investments and actions to improve the health of children and women for decades. And in the same way, good, reliable data are the foundation for understanding and tackling the problem of childhood lead exposure. These surveys underpin research that allows us to understand who is most affected and why. In India, for example, the 1998 DHS survey enabled rigorous and policy relevant analysis of the childhood correlates of blood lead levels in Delhi and Mumbai.
Beyond research, the experience of Georgia illustrates how real data can lead to real action. The Government of Georgia made the request to UNICEF to add a blood testing module in the upcoming MICS because it was concerned by reports of high blood lead levels in expats and lead levels in exported products. The 2018 MICS in Georgia tested 1,578 children aged 2-7 across the country, with samples subsequently sent to the Italian National Institute of Health for analysis. The results showed that 41 percent of Georgian children had elevated blood lead levels; in some regions, prevalence of lead poisoning was as high as 85 percent. With the results of the survey in hand, the Government of Georgia subsequently launched a national plan to identify sources of lead exposure and measures to manage the problem. Early indications suggest that the interventions following the MICS survey have reduced blood lead levels in children in Georgia.
The cost of (not) measuring childhood lead exposure in household surveys
What’s clear is that the cost of not measuring childhood lead exposure is unacceptably high. We need to get much, much better at measuring it. But that doesn’t mean it’s easy. Competition for inclusion of topics in DHS and MICS surveys is high. Measuring blood lead levels requires at minimum a finger prick test and so the inclusion of lead testing is high effort, when compared with other topics that may simply require interviews with households.
But the actual cost of including blood lead levels in household surveys is affordable, especially when compared with the cost of not testing and not confronting high levels of childhood lead exposure. In Georgia, where a nationally representative survey using “gold standard” venous testing took place, documentation shared by UNICEF suggests the cost was about $50 per test, for 2,600 tests. The rapid innovation of lateral flow and PCR tests for coronavirus demonstrate the cost reductions that come with scale. If there was mass demand for blood lead level testing across countries and regions, followed by investment in diagnostic innovation to allow for cheaper but reliable finger prick or dried blood testing, we would almost certainly see lower costs.
The case for measuring childhood lead exposure is clear
Nobody thinks it’s acceptable for millions of children to have their futures compromised by a dangerous neurotoxin that harms their health, development and life chances. And yet, for decades, we’ve collectively turned a blind eye to the pervasive lead poisoning across so many low and lower-middle income countries despite knowing the tremendous and irreversible damage it causes. That needs to change. Childhood lead poisoning is a problem that we can solve—and that needs to start with proper measurement.
Thank you to Abheet Solomon, Desiree Raquel Narvaez, Lyn Greer, Bo Pedersen, and Attila Hancioglu for helpful comments. All opinions, errors, and omissions are entirely our own. Thank you also to Aisha Ali and Rory Todd for excellent research assistance.
DISCLAIMER & PERMISSIONS
CGD's publications reflect the views of the authors, drawing on prior research and experience in their areas of expertise. CGD is a nonpartisan, independent organization and does not take institutional positions. You may use and disseminate CGD's publications under these conditions.