What if by taking a pill every day, all 33 million HIV-infected people in the world could not only fend off the deterioration of their own health, but also reduce their chances of infecting uninfected sex partners by 96 %? This is the prospect that is offered by newly announced results of the HPTN 052 trial. (See the Kaiser Foundation report
here, the UNAIDS announcement
here, the Global health Sushi report
here and the trial registry info
here. ) The trial has been cut short because only one among 877 HIV-infected people on anti-retroviral therapy (ART) infected his or her partner, while 27 among the 886 HIV-infected people did so. In view of the disadvantage apparently suffered by those taking the placebo instead of the real ART drugs, the researchers and their oversight board considered it unethical to continue to withhold the drugs from the couples in the control arm.
This dramatic evidence that ART lowers the infectivity of HIV supports the hypothesis of a beneficial biological external effect of ART. But is the evidence surprising and powerful enough to be a “game changer” as Michel Sidibe, the director of UNAIDS declared, or “to end, or at least diminish, a bitter feud within the AIDS world over how much funding should go to treatment versus prevention,” as the
Wall Street Journal has suggested?
It has long been known that ART can have both biological and behavioral effects on prevention. A simple typology of these effects classifies them as either beneficial (because they slow transmission) or adverse (because they speed transmission). (See the table from our 2004 book reproduced at the bottom of this blog.)
So the new experimental finding adds support to the upper left quadrant of this table positing a beneficial biological effect and thus to the proposition that I blogged in 2008
here and
here that these beneficial externalities of treatment might justify a greater allocation of resources towards treatment, as compared to a pure cost-effectiveness analysis that ignores such effects. In the majority of African countries that have not yet managed to treat a large proportion of those with more advanced AIDS disease, the push to use ART for prevention increases the tension between the ethical mandate to treat the sickest patients first and the stronger efficiency argument for allocating limited treatment resources to those who are not yet sick.
But it takes more than this confirmatory experimental trial to argue that prevention resources should be re-allocated towards treatment. Modeling shows that even if people start treatment when their CD4 count is 500, as they did in this trial, and then faithfully adhere to their daily dose for their entire lives, the future burden of the epidemic continues to rise until 2046. The two panels of the following figure project the future of the epidemic in Sub-Saharan Africa through 2050, incorporating the trial result that the infectiousness of treated persons is reduced by 96 %. These calculations also assume optimistically that financing will be found to enroll every HIV-infected person whose CD4 count is below 500, that circumcision roles out to 80 % of adult males by 2025 and that a 60% effective vaccine begins to roll out to all adults in 2025. Panel a of the figure shows that without enhanced behavioral prevention the number of Africans living with HIV/AIDS continues to increase until 2046, reaching a peak of 38 million before the AIDS transition occurs. And so does the cost of treatment, rising from its current level of around $6 billion to above $60 billion by 2050.
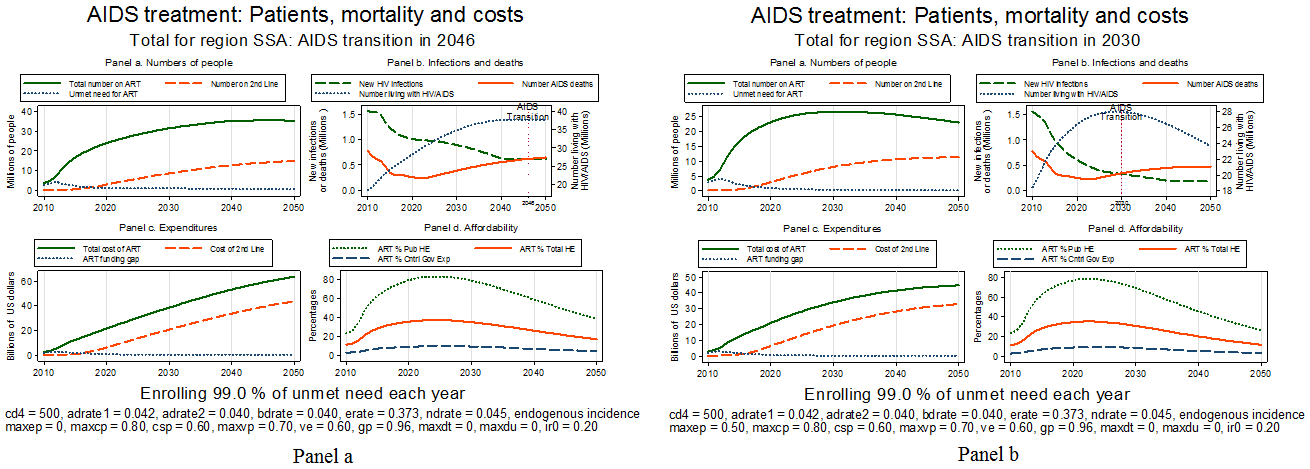
Figure. Simulations of the future African HIV/AIDS epidemic if treatment reduces infectivity by 96% and reaches everyone with a CD4 count below 500. Both scenarios assumed male circumcision reaches 80% by 2025 and a 60% effective vaccine begins to roll out in 2025. Panel a) assumes risk behavior is unchanged from current levels, while panel b) assumes a 50 % reduction in risk behavior.
(Source: Projections from the
AIDSCost model. If you own a copy of
Stata version 10 or later, you can produce the above projections by typing “findit AIDSCost” from inside Stata, installing the program, getting the data file and executing the following command for panel a:
“
aidsproj using aidscgd2010.dta, uptake(.99) takeoff(2010) horizon(2050) cd4(500) maxep(0) gp(.96) weo regions(SSA) graph grcombine” For panel b: Same command replacing
maxep(0) with
maxep(0.5). )
These projections assume constant cost per patient-year of both 1
st and 2
nd-line treatment, which seems reasonable since current pressures on unit costs are in both directions. Perhaps the unit cost of treatment will fall as pharmaceutical firms recognize the profit potential of 35 million customers at a low price as compared to 5 million at a high price. But no matter how inexpensive the individual drugs, the number of people taking them daily would still rise by a factor of seven, creating an ever larger dependency of recipient countries on the donors and pharmaceutical firms providing the drugs.
In contrast panel b shows a scenario combining 99% uptake at a CD4 level of 500 with effective behavioral prevention which reduces infections from high risk behavior by 50 %. Adding only this behavioral change moves forward the date of the AIDS transition by 16 years, to 2030, so that by 2050 the number of Africans living with HIV/AIDS has declined to 24 million and annual treatment expenditures will be about 25 % less than without this behavioral change.
With these considerations in mind, behavioral HIV prevention still deserves the highest priority. The potential savings in AIDS treatment cost from even small behavioral changes accumulate to extraordinarily large benefits over the next 40 years and effective prevention shields the African countries and their citizens from an important part of the looming burden of this disease.
 Source: Based on Table 3.3 on page 46 in Over et al., HIV/AIDS Treatment and Prevention in India: Modeling the Cost and Consequences (2004) available from Google Books here.
Source: Based on Table 3.3 on page 46 in Over et al., HIV/AIDS Treatment and Prevention in India: Modeling the Cost and Consequences (2004) available from Google Books here.

 Source: Based on Table 3.3 on page 46 in Over et al., HIV/AIDS Treatment and Prevention in India: Modeling the Cost and Consequences (2004) available from Google Books here.
Source: Based on Table 3.3 on page 46 in Over et al., HIV/AIDS Treatment and Prevention in India: Modeling the Cost and Consequences (2004) available from Google Books here.




