Recommended

Blog Post
From Deliberation to Design: NeoTest’s Second Working Group Meeting
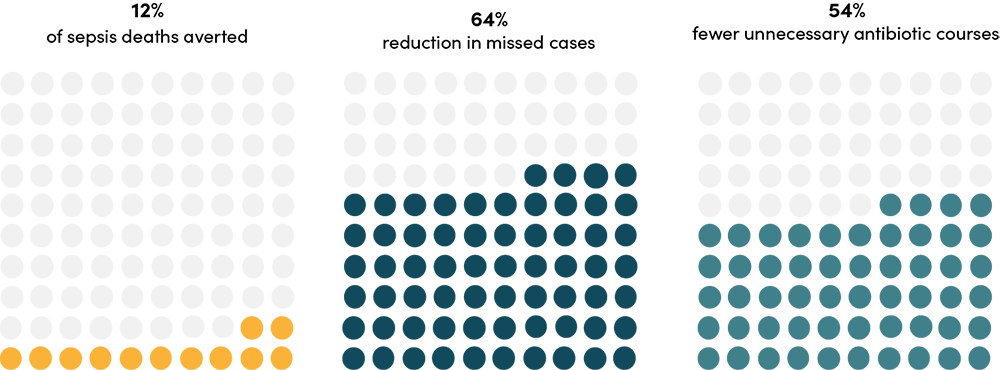
Key findings
A rapid, point-of-care diagnostic designed to rule neonatal sepsis in/out, would:
- avert more than half of unnecessary antibiotic prescribing, and
- reduce the number of cases that go undetected by almost two-thirds.
This would reduce mortality from neonatal sepsis by 12 percent and sepsis-related healthcare costs by 11 percent.
Every year, neonatal sepsis kills between 400,000 and 700,000 babies, overwhelmingly in low- and middle-income countries (LMIC). An estimated 84 percent of these deaths could be prevented with earlier diagnosis and appropriate treatment. Yet no point-of-care (POCT) diagnostic exists for this condition. The only available tool, blood cultures, takes 48 to 72 hours, far too slow to guide initial, life-saving decisions. As a result, clinicians rely on non-specific signs such as fever, lethargy, and poor feeding to decide whether to treat.
Figure 1. Modeled effect of a neonatal sepsis point-of-care test (POCT) on mortality and antibiotic prescription among infants with possible serious bacterial infections (PSBI)
Note: Possibly serious bacterial infection (PSBI) is a WHO-defined clinical syndrome in young infants (0–59 days) indicating potential sepsis, meningitis, or pneumonia, characterized by any of seven key signs: fast breathing ( 60/min), severe chest indrawing, fever (>38°C), hypothermia (<35.5°C), reduced movement, poor feeding, or convulsions.
In 2025, the World Health Organization (WHO) published a Target Product Profile (TPP) defining the minimum characteristics a rapid point-of-care triage test for neonatal sepsis should have. Building on prior modelling by FIND and Boston University, a new paper by HITAP, the National University of Singapore, and the Center for Global Development, with co-authors from WHO, AfroHTA, and HTA India, performed an early health technology assessment (HTA) of a hypothetical diagnostic meeting those criteria, estimating its potential health and economic impact.
What the study found
Using a decision-tree model validated by global experts, the study looked at the impact of a point of care test (POCT) meeting the WHO TPP, comparing it against current diagnostic and management pathways. This was done for two distinct populations of infants: infants managed in their facility of birth (“inborn") and infants presenting to community settings from home.
1. A diagnostic substantially increases the number of correctly diagnosed cases and reduces the number of missed sepsis cases
For every 1,000 inborn infants with suspected possible serious bacterial infection (PSBI), a test increased the number of correctly diagnosed cases (true positives) by 17 percent and reduced the number of missed cases (false negatives) by 62 percent. For the community cohort, these changes were 26 percent and 70.3 percent, respectively.
Figure 2. Modeled effect of a neonatal sepsis POCT on diagnostic outcomes across a cohort of 2,000 infants with possibly serious bacterial infection
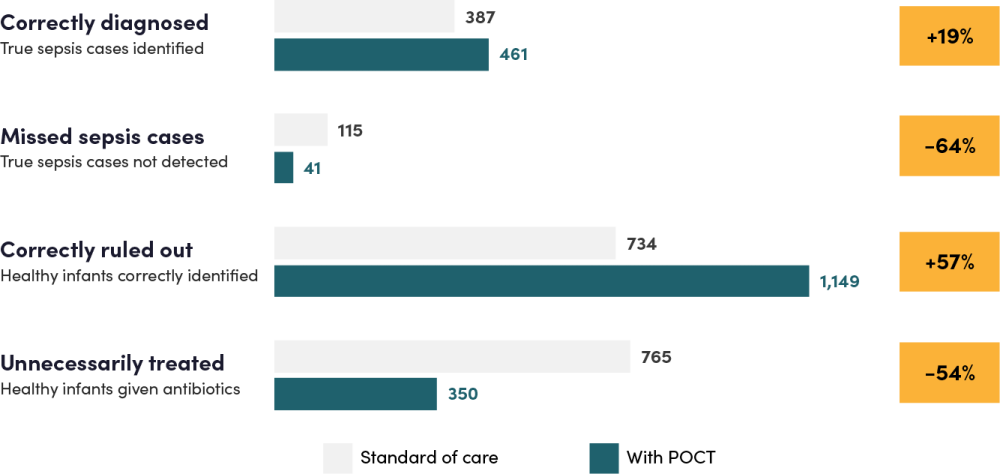
Note: The cohort of 2,000 infants with PSBI is split between 1,000 infants from the inborn arm and 1,000 infants from the community arm. Estimated numbers represent the sum of results across the two arms of the model. “Correctly diagnosed” and “missed sepsis” refer to true-positive and false-negative classifications; “correctly ruled out” and “unnecessarily treated” refer to true-negative and false-positive classifications. Possibly serious bacterial infection (PSBI) is a WHO-defined clinical syndrome in young infants (0–59 days) indicating potential sepsis, meningitis, or pneumonia, when any one or more of the following signs is present: fast breathing (60/min), severe chest indrawing, fever (≥38°C), hypothermia (<35.5°C), reduced movement, poor feeding, or convulsions.
2. The health and economic benefits of more accurately identifying which babies have sepsis, and which do not, are significant
The direct effect on mortality was substantial—deaths from neonatal sepsis fell by 13.3 percent in the inborn cohort, and 9.1 percent in the community cohort.
The test’s value extended beyond its diagnostic function. In the inborn cohort, correctly ruling out non-sepsis cases eliminated nearly 1,000 unnecessary inpatient bed-days per 1,000 infants. Shorter hospitalization, in turn, reduced exposure to hospital-acquired infections (HAIs)—a major driver of neonatal mortality in under-resourced wards—resulting in a 20 percent reduction in HAI deaths.
Table 1. Modeled effect of a neonatal sepsis POCT on mortality, DALYs, total inpatient days, and total healthcare costs across a cohort of 2,000 infants with possibly serious bacterial infection
| Standard of Care | With Point-of-Care Test (POCT) | Percentage change | |
|---|---|---|---|
| Health and economic outcomes | |||
| Deaths from neonatal sepsis | 78 | 69 | -12% |
| DALYs from all causes | 3,073 | 2,740 | -11% |
| Inpatient hospital days | 8,821 | 7,612 | -14% |
| Healthcare costs | $383,347 | $340,291 | -11% |
Note: The cohort of 2,000 infants with PSBI is split between 1,000 infants from the inborn arm and 1,000 infants from the community arm. Estimated numbers represent the sum of results across the two arms of the model. Possibly serious bacterial infection is a WHO-defined clinical syndrome in young infants (0–59 days) indicating potential sepsis, meningitis, or pneumonia, when any one or more of the following signs is present: fast breathing (60/min), severe chest indrawing, fever (≥38°C), hypothermia (<35.5°C), reduced movement, poor feeding, or convulsions. DALY = disability adjusted life year.
3. When quantified in DALYs and dollars saved, the impact of the test is profound
Quantified in disability-adjusted life years (DALYs) and dollars, the test would save 206 DALYs and over US$37,000 in healthcare costs per 1,000 inborn babies; for community-presenting babies, 127 DALYs and nearly US$6,000. The majority of the DALY gains are driven by reductions in the number of neonates dying from sepsis (81 percent).
DALY gains also come from two sources that most cost-effectiveness studies omit: reduced antimicrobial resistance (AMR) from fewer unnecessary antibiotic prescriptions, and avoided long-term complications, like necrotizing enterocolitis and inflammatory bowel disease linked to early life antibiotic exposure. We find a reduction in AMR-attributable DALYs of 22 percent in the inborn cohort and 35 percent in the community cohort, and reductions in complication-related DALYs of 14 percent and 43 percent, respectively.
4. These results held up under extensive sensitivity analyses
Even when key parameters were varied (such as the baseline prevalence of neonatal sepsis, how unwell babies are when they first present), the test was the dominant strategy (being both clinically superior and cost saving) in 93 percent of simulations for the inborn cohort and 85 percent for the community cohort, with a maximum value-based price of over US$350 for inborn settings and US$200 in community settings.
Figure 3. The POCT dominates standard of care: improving outcomes while reducing costs
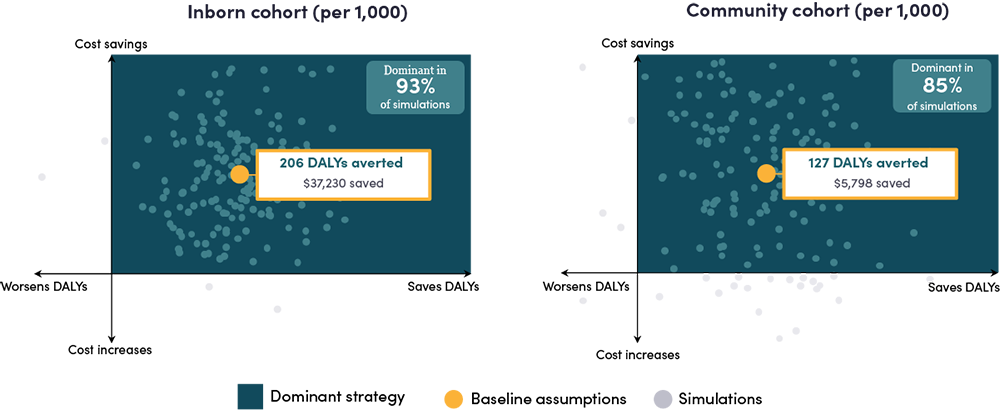
Note: Each dot represents one simulation varying all model parameters simultaneously. The base case and the vast majority of simulations fall in the dominant quadrant (green): better outcomes and lower costs. "Dominant" = POCT is simultaneously more effective and less costly than SOC. Simulation spread is illustrative of the probabilistic sensitivity analysis (10,000 Monte Carlo iterations).
Why this matters
The paper shows that a test meeting WHO TPP specifications would be dominant—simultaneously improving health outcomes and reducing costs—across diverse LMIC settings. Even under conservative assumptions, the test’s value-based price exceeds US$200 per unit, compared to a target price of US$5.
Yet no such test exists. This is not because it is technically impossible—it is because it is commercially unviable. Diagnostic developers face fragmented LMIC procurement, uncertain demand, and a reimbursement landscape that does not reward innovation. The result is a product that would pay for itself many times over, but that no firm has sufficient incentive to develop.
The paper provides the missing evidence base for donors and policymakers to build a procurement and reimbursement case. It quantifies the return: for every 2,000 neonates tested, we would save nine lives and avert over $43,000 in healthcare costs. And for the global health community, it reinforces that accelerating diagnostic innovation for neonatal sepsis is a high-value investment towards reducing newborn mortality.
DISCLAIMER & PERMISSIONS
CGD's publications reflect the views of the authors, drawing on prior research and experience in their areas of expertise. CGD is a nonpartisan, independent organization and does not take institutional positions. You may use and disseminate CGD's publications under these conditions.
Thumbnail image by: Adobe Stock Images