This blog is part of a series by CGD ahead of the EU-Africa Summit which will begin on 17th February 2022. This series presents proposals for priorities, and commentary on whether a meaningful reconstruction of the relationship between the two continents is likely.
Last week, the European Union and its member states placed a “European global health strategy” at the top of their shared agenda. And as part of the EU-Africa summit this week in Brussels, the EU is highlighting its support for regional manufacturing of COVID-19 vaccine via the African Union’s Partnership for African Vaccine Manufacturing (PAVM) as a major piece of the overall agenda. Sub-Saharan African countries currently import 99 percent of their vaccines, and the EU seem to be acknowledging that ensuring that Africa has autonomy over its own health security is important, announcing over €1 billion in cash plus in-kind expertise for PAVM, with more on the way.
To date, much of the discussion has focused on the EU’s supply side support as well as disagreements between the AU and the EU on intellectual property around the mRNA vaccines. But there are also major uncertainties ahead that all parties must understand to determine a joint way forward: How much financing is required to support regional manufacturing on the demand side, meaning how much vaccine would need to be procured for new manufacturers to survive and thrive, and over what period? What kind of vaccine manufacturing makes economic sense for the medium-term? Who will finance and procure doses from regional manufacturers? Answering these questions and planning ahead is necessary for domestic manufacturing of vaccines in Africa to become a successful reality. We look at three sources of demand-side uncertainty and call on the EU to provide significant and long-term demand-side financing that can deliver success.
Uncertainty #1: Will there be future demand for COVID-19 vaccine in sub-Saharan Africa?
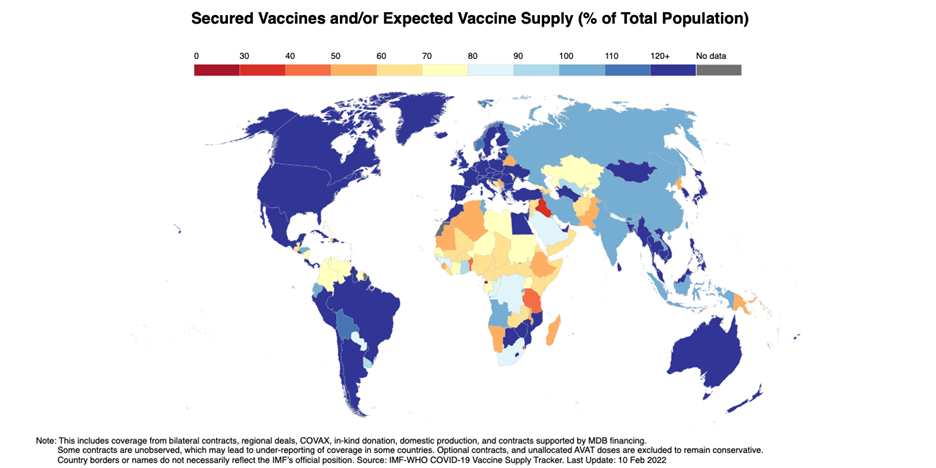
Most countries in sub-Saharan Africa have procured or secured COVID-19 vaccine sufficient to cover at least half of their total population with two doses. While a new and more virulent variant is a future possibility, WHO AFRO last week announced their view that the region was transitioning out of the pandemic phase of the disease given the extent of population immunity. The week before that, a group of prominent South African health experts noted that the goal of vaccination was no longer to reduce transmission but to protect vulnerable groups, a strategy that might require a lower number of doses (and a lower coverage target) than the amount already ordered or secured via donation.
Also affecting future demand is the unanswered question of whether annual or repeat booster vaccination against COVID-19 will be required; there is considerable uncertainty on the answer to this question, but it is not a foregone conclusion that repeat doses are needed. Further, the WHO has pointed out that repeat doses of current COVID vaccine may be an unsustainable strategy. And future demand for COVID vaccines may shift to multivalent vaccines containing antigens from different SARS-CoV-2 variants of concern.
Uncertainty #2: Is the global vaccine market already saturated?
There is also the question of the now-huge global supply of vaccines. As of February 10th, the IMF-WHO COVID-19 Vaccine Tracker finds countries had cumulatively ordered about 38.7 billion doses from existing manufacturers. Countries have ordered so much vaccine that major player AstraZeneca assumes that their market from COVID-19 products will decline this year, in 2022.
Further, new vaccines —potentially more cost-effective— are coming online. While South Africa’s Afrigen recently announced its reverse engineering of Moderna’s mRNA and formulation with guidance from WHO, the Medicines Patent Pool, the South Africa Medical Research Council, and the Africa Centres for Disease Control, the current version of the vaccine is difficult for many countries to deliver. Some have pointed out the “newer, shinier” mRNA vaccines may have distracted from vaccine candidates like Novavax that “has roughly the same efficacy as the mRNA vaccines with the added benefit of being based on an older, more familiar science.” Afrigen aims to innovate to lower the cost and ease delivery constraints of the Moderna mRNA vaccine, which could be good for potential uses of mRNA vaccines for other diseases, but it’s not clear that a government in sub-Saharan Africa would seek to adopt and finance more mRNA vaccine for COVID in lieu of a vaccine like Novavax, already being produced at multiple large scale manufacturing sites. Add to the mix new vaccines like the patent-free Corbevax, it is simply not clear that mRNA manufacturing is the only future direction of travel in response to COVID-19 at least, though mRNA has many potential applications in terms of vaccines for other infectious diseases and possible benefits of speed in developing and manufacturing strain matched vaccines for future SARS-CoV-2 variants of concern.
Shortages of critical input materials for vaccine manufacturing such as single use bioreactors, reagents and assays in the last 12 months has also made it clear that setting up vaccine manufacturing plants without adequate foresight into sourcing and the overall supply eco-system may not necessarily lead to greater supply security or supply chain resilience.
Finally, as a recent BMJ piece notes, both Moderna and BioNTech have announced their own initiatives to build vaccine plants in Africa. Moderna said it planned to spend €437 million to make 500 million doses of mRNA vaccine each year beginning in 2023, while BioNTech planned to begin plant construction in mid-2022 for 50 million doses of vaccine per year.
At minimum, these circumstances mean that it will be difficult for new regional manufacturing entrants to join the market unless they are highly competitive on price or there is a large subsidy. So, the question for the EU is — will the EU put up the amount of subsidy needed for success? What is this amount? And given the competitive landscape and the EU’s reluctance to support a patent waiver, would money to procure a guaranteed annual number of regionally produced doses via the African Union be a more useful tool to create the conditions for at least some of the entrants to succeed, gain efficiencies, and stay in the market?
Uncertainty #3: Who will finance demand, and what are the roles of the different procurers?
Pre-COVID, aid was still financing much of vaccine purchase in sub-Saharan Africa with the exception of South Africa. Some of this expenditure was modestly co-financed by domestic resources via Gavi (though transition away from Gavi aid and towards domestic finance was slated for many of the larger middle-incomes in the region from 2023: Nigeria, DR Congo, Ethiopia, Tanzania, Cote d’Ivoire, and Mozambique). During COVID-19, some countries exceptionally borrowed from the World Bank or used domestic revenues to finance a share of the AU African Vaccines Acquisition Task Force (AVATT) purchase of COVID-19 vaccine. Now, countries are struggling to recover from the COVID-19 shock and declines in public revenues alongside increasing levels of unsustainable debt. Together, this may mean that spending more on vaccines —not just for COVID-19 but for all vaccine-preventable diseases— may be a very heavy lift from domestic financing.
The question then is — who will finance the necessary subsidies for regional manufacturing firms to grow and prosper? Could the EU have a major role in this financing? If more domestic spend is envisioned, what are the trade-offs in spending by governments as basic needs in the health sector remain unmet? Will government subsidies for regional manufacturing firms require a change in procurement policy via Gavi to source a minimum amount, or an approach where pooled procurement moves increasingly to the AU level and Gavi’s traditional donors like the EU move their money to the AU’s AVATT? Defining the way forward will be necessary to construct the kinds of forward-looking guarantees and contracts necessary for regional manufacturing to survive, and it is unlikely that AU governments themselves will be providing all or even most of the financing required, barring major reallocations from other uses.
What to do in the next phase of the EU-AU partnership
As AU and EU leaders meet this week, it is time to consider the medium-term trajectory. While most of the energy so far has gone towards supply side subsidies for vaccine development and manufacturing, and the disputes around intellectual property and patent waivers, it is important to take a more holistic view and assess what measures can balance the twin objectives of supply security and ability to procure and finance cost-effective vaccines at affordable prices. Even supply side subsidies need to emphasize flexibility in vaccine manufacturing in order to cope with some of the uncertainties highlighted.
There are of course many motivations to support vaccine manufacturing closer to home, including security, supply chain resilience, and knock-on effects for tertiary education and scientific innovation, but the AU and EU should work to develop a clearer picture on the demand side, and put large and long-term financial support from the EU in place to support. These key actions will help the AU’s PAVM initiative achieve its intended impact on population health.
DISCLAIMER & PERMISSIONS
CGD's publications reflect the views of the authors, drawing on prior research and experience in their areas of expertise. CGD is a nonpartisan, independent organization and does not take institutional positions. You may use and disseminate CGD's publications under these conditions.