Recommended
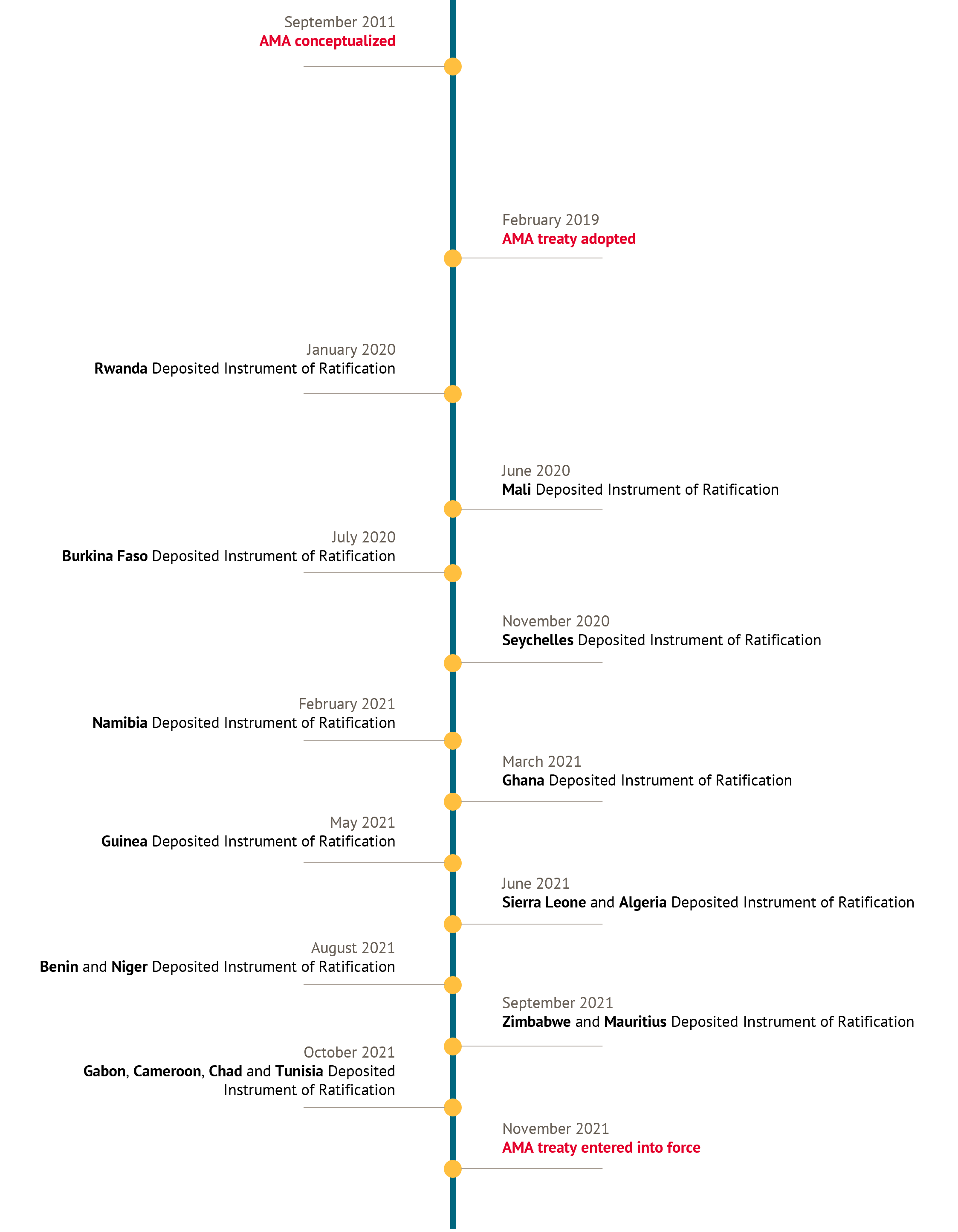
As of October 5, 2021, 15 African countries ratified the African Medicines Agency (AMA) treaty, entering into force the second specialized health agency of the African Union. The treaty struggled to get sufficient support for more than a decade after AMA was conceptualized at the 60th World Health Organizational Regional Committee for Africa session and more than two years after its adoption in February 2019. However, the initiative recently gained momentum likely due to a combination of efforts, including the appointment of a Special Envoy to help garner support for the treaty, the push to deliver on the Partnerships for African Vaccine Manufacturing, and the success of the other specialized health agency of the African Union—the Africa Centres for Disease Control (Africa CDC). As a result, five of the now 17 countries deposited their ratification within the last six months, and most African Union member countries have now ratified or signed it.
Similar to Africa CDC, AMA will be a continental platform with wide-reaching influence. AMA also builds on the encouraging experience from previous regional regulatory partnerships in Africa, especially those in Regional Economic Communities (RECs) such as the East African Community (EAC) and the Southern African Development Community (SADC). In line with these other efforts, AMA is positioned to improve regulatory harmonization and reliance, increase efficiency and effectiveness, and unify the approach to product reviews and approvals across the region, which will facilitate quick and reliable access to safe, effective, and quality-assured medicines—a key component of Universal Health Coverage.
Limited resources contribute to poor regulatory outcomes across the continent. AMA can help Africa leapfrog from low existing levels of regulatory capacity to a regional harmonized approach.
Limited resources contribute to poor regulatory outcomes across the continent. AMA can help Africa leapfrog from low existing levels of regulatory capacity to a regional harmonized approach. The World Health Organization estimated that only seven percent of African countries had moderate capacity for medicines regulation and more than 90 percent had minimal to no capacity. General resource shortages also contribute to the highest regional prevalence of substandard and falsified medicines and reported delays of four to seven years between the submission of a new product to a regulatory agency in a high-income country to final approve in sub-Saharan Africa. By supporting capacity building and harmonizing standards and processes, AMA can help strengthen medicines regulation amidst resource constraints.
AMA’s ability to deliver on these objectives depends on key decisions made at this initial stage regarding the structure and initial priorities of the agency. With sufficient political support for AMA now secured, stakeholders are shifting their focus to questions of strategic design and operationalization. One such meeting is taking place today, as the African Medicines Regulatory Harmonization (AMRH) Partnership Platform convenes to establish strong collaboration and communication among members from the start. This blog weighs in on these discussions, highlighting three foundational considerations to factor into the agency’s structure and plans to ensure its sustainability and success.
1. AMA must clearly establish its priorities and its relationship to other national, regional, and international entities and initiatives.
AMA must clearly decide on the regulatory functions it will prioritize first (e.g., marketing authorization vs pharmacovigilance vs clinical trial oversight), how its work will leverage the existing capacity on the continent while avoiding duplication of efforts, and the best way to translate its recommendations into regulatory decisions at the national level. These decisions will not be made in a vacuum, as AMA is entering a space already crowded with several national, regional, continental, and global initiatives and networks with different degrees of success and political buy-in. For example, some countries like Ghana and Tanzania already have functional National Regulatory Authorities that can carry out all regulatory activities. Other countries like Rwanda and South Africa are fast tracking regulatory strengthening efforts to bolster local manufacturing capacities. Several other countries have neither the capacity nor the interest to invest in more robust regulatory systems.
The situation at the regional level is similar. A few RECs, such as EAC and SADC, have made substantial progress by harmonizing procedures and conducting joint reviews of new medicines coming to their markets. Other RECs, such as the Community of Sahel–Saharan States and the Arab Maghreb Union, have not prioritized medicines regulation and have made little progress.
Finally, continental regulatory initiatives such as the African Vaccines Regulatory Forum and global initiatives such as the WHO collaborative procedure for accelerated registration have provided useful platforms for collaborative work even though it has been challenging to translate joint assessments into decisions at the national level.
AMA should be strategic and focus on areas where the biggest results can be obtained (e.g., harmonizing procedures to conduct and recognize good manufacturing practices from functional authorities, joint assessments to support the marketing authorization of essential products like oral antivirals to treat COVID-19, and post marketing surveillance and quality control of tracer medicines). AMA must do so by determining the optimal set of functions to conduct at the national, regional, and continental levels. These decisions will inform the amount and type of resourcing needed by the agency (see point 2 below).
2. AMA must be independent and financially viable.
AMA will need to establish an adequate legal, institutional, and operational framework to secure its autonomy from the African Union and remain free from political influence. This independence, which will guarantee its credibility for any regulatory decisions, must be built into the legal framework for AMA, establishing a governance arrangement that allows the agency to develop its own financing channels and project management capabilities. As a comparable agency subsumed by the African Union, the governance arrangements of Africa CDC can serve as a valuable model.
Decisions on the key regulatory functions to be conducted at the regional and continental level and prioritized in the first years will determine the budget and staffing AMA requires. These resources must be secured. Regulatory authorities are chronically under-resourced, especially in low-and middle-income countries where there is high staff turnover and limited availability of regulatory professionals. Unless AMA is staffed and financed adequately, it risks facing similar challenges on a larger scale.
A key challenge for AMA will be to secure long-term, sustainable funding. As important as the amount of financing required for effective and sustainable functioning is the structure for financing AMA. A critical part of establishing AMA’s independence is developing sufficient financial autonomy, meaning its budget must comprise of direct support from the African Union as well as fees charged to industry. The industry fee arrangement will need to be decided through discussions with manufacturers on the appropriate level and timing of fees.
In the meantime, short to mid-term support from bilateral donors—including high-income country governments, philanthropies, development banks, and global procurement agencies—is necessary to ensure AMA can function effectively. Donors can revitalize existing funding streams to support this new agency. For example, the World Bank established the Global Medicines Regulatory Harmonization Multi-Donor Trust Fund in 2011 with an initial contribution of $12.5 million from the Bill and Melinda Gates Foundation. Additional funds should be invested into funds like this to provide adequate financing as AMA promotes harmonization of medicines regulation and strengthens regulatory systems across the continent.
3. AMA must support regulatory capacity building at the continental, regional, and national levels.
The continental focus of AMA should not shift resources and attention entirely away from the regional and/or national level. Rather, AMA will be well positioned to support capacity building in a pragmatic strategy of learning-by-doing at the regional and national levels.
AMA should learn from what has worked and has not worked with the Regional Centres for Regulatory Excellence (RCOREs) – an initiative of the AMRH Program within the African Union Development Agency’s New Partnership for Development. Founded in 2009, AMRH has served as a continental platform to expedite effective regulatory processes for quality-assured medical products in African markets. AMRH recognized a general lack of regulatory expertise on the continent and, in response, established 11 RCOREs that offer training to strengthen regulatory capacity in quality assurance and quality control; medicines registration and evaluation; licensing of the manufacture, import, export, and distribution of medical products; inspection and surveillance; and clinical trials oversight. RCOREs have successfully increased the technical capacity of African regulators with targeted curricula and trainings, slowed the “brain drain” of regulatory expertise, and supported peer-to-peer learning. However, RCOREs struggle to secure adequate and sustainable financing and often miss opportunities to prioritize regional over national work and communicate lessons from their activities for continental and global learning. Not only can AMRH serve as the foundation for a regional platform for regulatory harmonization in Africa, but AMA can adapt its RCORE model as a cost-effective strategy for capacity building at the national level.
Building on the RCORE example, we propose an AMA Academy in partnership with the World Health Organization Academy recently launched in France. AMA Academy could leverage expertise from across the African continent to offer a standard curriculum for national regulatory authorities, increasing technical capacity on core regulatory functions. Strengthening medicines regulatory science expertise at all levels is a key step towards strengthening the broader network of regulators that will produce and implement regulatory decisions.
Conclusion
The strategic decisions made in these early days will critically determine AMA’s longevity, legitimacy, and effectiveness. Decision-makers in Africa must move quickly to set regulatory priorities, secure adequate initial financing with support from donors, and establish a strong governing framework to start AMA on solid footing. At the same time, they must also draw up plans for sustainable financing and capacity building activities to ensure AMA continues to function effectively and strengthens the regulatory system in which it operates. African policy makers and donors must seize this unique opportunity to design and operationalize AMA for greatest impact. Join us for a virtual event on February 24 from 9:30-11:00am ET to continue the discussion.
Topics
DISCLAIMER & PERMISSIONS
CGD's publications reflect the views of the authors, drawing on prior research and experience in their areas of expertise. CGD is a nonpartisan, independent organization and does not take institutional positions. You may use and disseminate CGD's publications under these conditions.
Thumbnail image by: Adobe Stock