In April this year, the National Academies of Science, Engineering, and Medicine (NASEM) published a report making the case for “Integrating Clinical Research into Epidemic Response.” As reflected in its title, the 250-page-plus-appendices report makes a strong evidence-informed argument for integrating health service delivery with clinical research conducted during epidemics. The goal is to produce critical information on the efficacy and safety of potential therapeutics and vaccines for tackling such epidemics after they occur, or, better still, for preventing them from happening. Earlier this week, the group reconvened at the Wellcome Trust to discuss “what next.” The need to focus on systematic support and funding for the data collection and research functions in outbreak-affected countries came out again as the top priority.
Ebola might be over for now, but there is no such thing as an outbreak-free period. Last week’s WHO Bulletin on Outbreaks and Other Emergencies flags lethal outbreaks of cholera in the DRC, Nigeria, South Sudan, Angola, and Tanzania; an outbreak of monkeypox in northern Nigeria; the plague in Madagascar; and Lassa fever in Nigeria—plus a most deadly and fast-spreading cholera outbreak in war-torn Yemen. But for a disease like Ebola, where no vaccine or therapeutic product currently exists, and where the only way such a vaccine or treatment can be tested and licensed for use in humans is through research carried out in the context of the next outbreak, the message of the NASEM report is a critical one: research saves lives. (The NASEM report findings were discussed a few weeks ago at CGD. A video of the event and the presentations are available here, and my colleagues Mead Over and Roxanne Oroxom wrote a blog about the major findings and recommendations.)
Here are some of the takeaways coupled with a few thoughts and suggestions:
-
According to the report, building research capacity integrated with service delivery (i.e., ranging from routinely collected data/registers to clinical trial capabilities across all providers rather than higher-tier university ones alone) is a prerequisite for preventing/addressing the next big outbreak. But what does “integrated research capacity” look like in countries with extremely weak routine administrative data to start with, underperforming national statistics systems, and unreliable or nonexistent healthcare provider claims data to drive reimbursement even for everyday procedures? Is it realistic to aspire to a US-like learning healthcare system model (see here for a discussion of what this would look like in an African setting)? Further, “clinical research capacity” (even in the narrowest sense of being able to collect and analyse routine epidemiological surveillance data) does not currently form part of the International Health Regulations which 196 countries have signed up to and which is meant to safeguard global public health against outbreaks. Should IHR be revised to include “research capabilities”? And if this is bureaucratically impossible, how can the profile of research/data collection as a core element of good quality, affordable, and equitable care provision be raised and countries held to account for making a policy and financial commitment towards research infrastructure? Could financial incentives help move this agenda in the right direction?
-
Who pays for it all? On-going, adequate, and stable funding for data/surveillance improvements and research capacity (as opposed to project or outbreak-specific) is not currently available. In a report on Data for African Development, the African Population and Health Research Center and CGD proposed a data compact to help mobilise and focus external and domestic resources towards progress on national statistical priorities. Without accurate, accessible, timely and unbiased data on basic things such as births and deaths, even deriving incidence rates becomes an impossible task.
-
Should African governments be expected to foot the bill? Contribute, including through streamlining regulations, maybe. But for many sub-Saharan African countries with competing demands on their small healthcare budgets and little appetite on the part of national treasuries to prioritise health over other public spending, spending on research infrastructure is a tall order (figure 1 in WHO’s Public Financing for Health in Africa report shows that for more than half of the African countries studied, healthcare spending was reduced or stayed roughly the same in recent years). Also, this is not only an African problem; the results of such research are a regional or even global public good.
-
Should aid money be allocated towards building research capabilities? Such capabilities could qualify as global public goods (GPG), and many have called for international financing on these grounds (CGD’s Nancy Birdsall and Anna Diofasi make a case here for more investment by bi- and multilaterals in development-related GPGs through an ODA-plus approach). But while calls to do more abound, progress in allocation is minimal (or modest), with about a fifth of total ODA going towards GPGs.
-
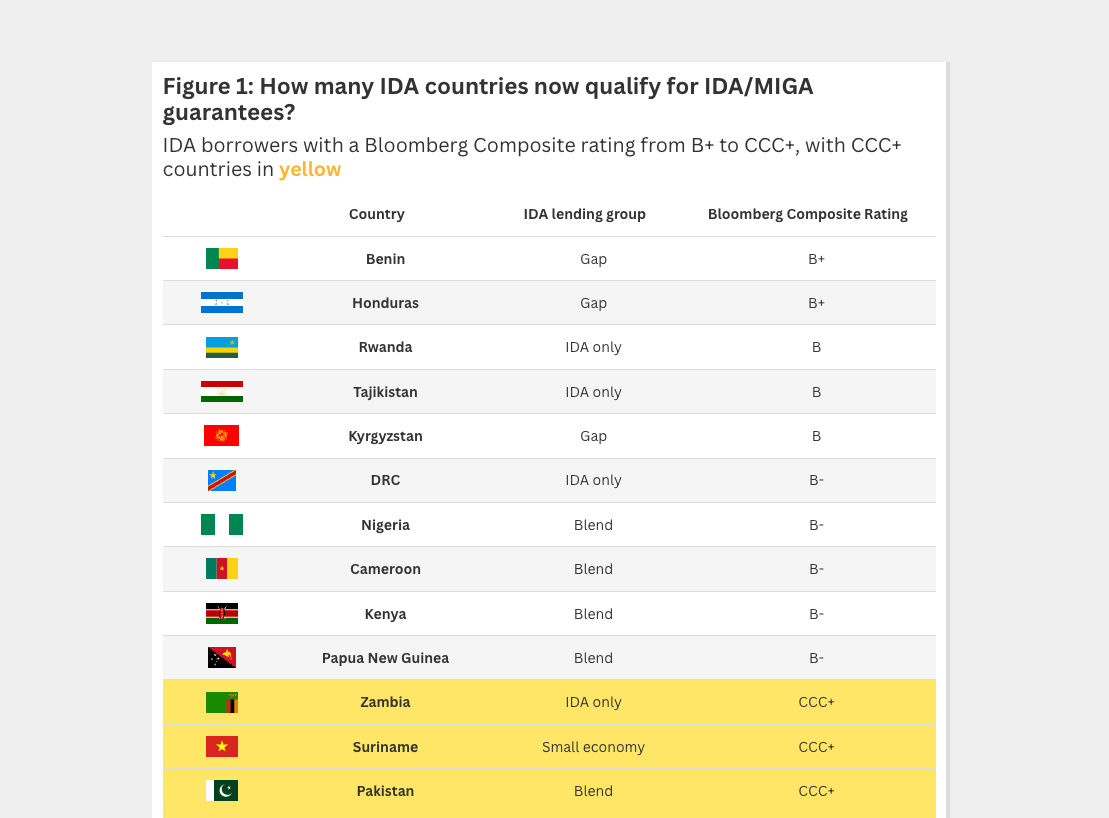
A practical approach, with synergies with the equally pressing anti-microbial resistance (AMR) action agenda, has been proposed by Glassman here. The International Development Association’s (IDA) regional programme (less than 3 percent of which currently goes towards public health strengthening) can be used for strengthening very weak public health systems, including preparedness and surveillance as well as research capacity for doing research during outbreaks, so that effective vaccines and treatments can be tested and rolled out. A small fraction of the commitment can be linked to results. Indicators can include “research capacity” or, better even, proof of an effective research function, focusing governments’ mind on getting the job of data collection and synthesis done rather than counting the number of African PhDs graduating from eminent US and EU universities. Payment can also be linked to things like lab readiness (though high-tech equipment alone won’t do the trick); functional engagement between governments and their academic centres, with established career pathways for local researchers committed to working on policy; and, drawing on social sciences, proxies for “trusted institutions” (see here and here for some preliminary findings from Liberia and Sierra Leone). All of the above would carry positive spillovers on health system strengthening in between outbreaks. Linking to results can spur demand on the government side. Without such a nudge, there is a risk that governments go for high-tech equipment more than systems, with the World Bank and regional development banks serving as procurement agents on behalf of senior government officials. The above can work in tandem with the World Bank’s recently launched Pandemic Emergency Financing Facility (PEF). Though explicitly excluding investments in health system strengthening in between outbreaks (assuming such in-between periods actually exist), PEF can be used to fund clinical research and data collection during outbreaks as part of the emergency response.
-
Should African governments be expected to foot the bill? Contribute, including through streamlining regulations, maybe. But for many sub-Saharan African countries with competing demands on their small healthcare budgets and little appetite on the part of national treasuries to prioritise health over other public spending, spending on research infrastructure is a tall order (figure 1 in WHO’s Public Financing for Health in Africa report shows that for more than half of the African countries studied, healthcare spending was reduced or stayed roughly the same in recent years). Also, this is not only an African problem; the results of such research are a regional or even global public good.
-
For it to be policy-relevant, research must be pragmatic: it must reflect the realities (including budgetary, human resource-related, and other constraints) of the healthcare systems whose problems it is meant to address. Developed economies have realised this and have established pragmatic, patient-centred knowledge-generation systems (including earmarked funding for research and capacity building) such as the National Institute for Health Research in the UK and the Patient Centered Outcomes Research Institute in the US. Further, groups of countries, such as the EU, have come together to invest in cross-border research partnerships such as PREPARE, specifically aimed at responding to outbreaks and “providing real-time evidence for clinical management of patients and for informing public health responses.” No such initiative yet exists in sub-Saharan Africa. The European & Developing Countries Clinical Trials Partnership, on the other hand, is increasingly committing funds to building research capacity for research during outbreaks, though its focus is still on efficacy more than effectiveness.
-
The UK’s DFID and other ODA research monies can potentially add value here, especially if funds can be released not for doing research, but for building capacity to do research (e.g., streamlining regulations and ethics approvals processes, and strengthening IT and data collection systems) in-country. But for this to happen, as highlighted in the UK aid’s watchdog review of ODA-funded research, there may have to be better top-down targeting of significant investment in potentially high-value areas. DFID’s latest call for proposals in this space—the Tackling Deadly Diseases in Africa Programme—is a good start, but it remains to be seen whether it ends up suffering from too diffuse a vision and too little money per consortium to make a difference. Consistent with its mission, the National Security Council could, through the UK’s Conflict, Stability and Security Fund (currently under review), take on the task of implementing the NASEM report and, in doing so, crowd in funds from diverse sources, such as DFID’s CDC and other major players in this space, like the Wellcome Trust. As the Independent Commission for Aid Impact called for, such an investment would define UK aid’s legacy in this space for years to come, and could be a global security and stability intervention that the UK government could proudly advertise rather than conceal.
- Coordination, governance, and politics. There is a plethora of initiatives, networks, and networks of networks of researchers and research projects across sub-Saharan Africa. Some have been around for quite a while and some came about after the Ebola tragedy—WATER, ALERRT, the Ebola response anthropology platform, CREDO, ISARIC, to name but a few (the WHO is apparently working on mapping them out). But they (at least the Ebola-related ones) tend to be ad hoc and to rely on fragmented, short-term donor funding for their sustainability. There is perhaps a case to be made for these to be better coordinated and for longer-term funding to be sourced to ensure they are sustained beyond the next outbreak. But it is so far not clear who is willing and has the legitimacy to lead in a coordinating and fundraising/pooling role. In a recent Lancet comment, the NASEM report authors call for better and better-distributed governance roles to avoid the kind of conflicts of interest observed during the previous outbreak (see here for a great conflict-of-interest matrix). But if not WHO, then who? This is still work in progress and the topic of the next meeting, it seems.
DISCLAIMER & PERMISSIONS
CGD's publications reflect the views of the authors, drawing on prior research and experience in their areas of expertise. CGD is a nonpartisan, independent organization and does not take institutional positions. You may use and disseminate CGD's publications under these conditions.
Thumbnail image by: Social media image by DFID