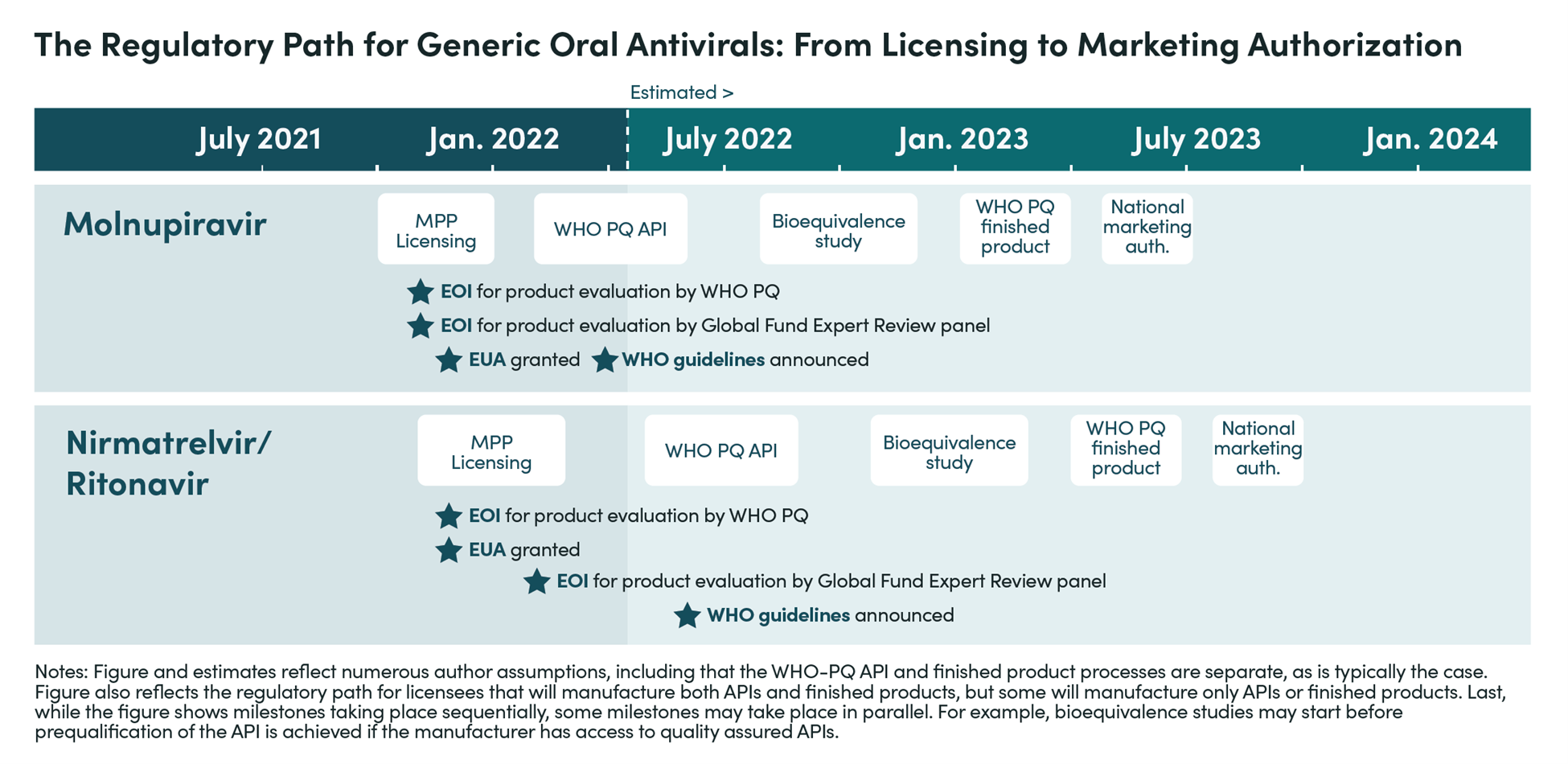
In December 2021, two treatments for COVID-19—Paxlovid, developed by Pfizer, and Lageviro, developed by Merck—received emergency use authorization in the US. One year later, access remains concentrated in wealthier countries and demand in lower-income countries is extremely low. Pfizer and Merck signed voluntary licensing agreements with select manufacturers to produce generic versions at more affordable prices, but these products are not expected to be available until 2023. Even then, insufficient deployment infrastructure may hamper patient access.
As COVID-19 evolves into an endemic disease and macroeconomic challenges squeeze health budgets, we need a more realistic strategy. Governments and donor partners must advance a more strategic approach with targeted goals for oral antiviral access. In the near-term, efforts to stabilize demand, speed up the production of high-quality generics, and lay the infrastructure for delivery will serve as an important insurance policy if—or when—a dangerous new variant emerges, especially in settings where vaccination coverage is low. In the longer-term, the global community must learn from this experience to accelerate access to oral antivirals and other medical countermeasures to combat future disease outbreaks.
A new body of CGD research examines the current state of play on COVID-19 antivirals. They provide a deep dive into key cross-cutting areas—demand, voluntary licensing for generic supply, and deployment—and offer policy actions for 2023 and beyond.
Access to COVID-19 Antivirals and Future Outbreak Medical Counter-Measures
More from the Series
Blog Post
December 14, 2022
Four new CGD pieces, released today, examine the current state of play on COVID-19 oral antivirals. Our analyses provide a deep-dive in key cross-cutting areas—demand, voluntary licensing deals for generic supply, and deployment—and offer policy actions for 2023 and beyond.
CGD NOTE
May 11, 2022
This note takes a quick look at the lessons learned and the existing landscape of MCM manufacturing in the context of the current pandemic response and suggests eight areas for action along with near-term recommendations to the global community to both prepare and respond to future pandemic risks.
CGD NOTE
April 07, 2022
While oral antivirals are not a substitute for vaccination, they are key to preventing severe illness, saving lives, and preserving health systems. Vaccination rates continue to be low in low-income countries; only 14 percent of the population in low-income countries has received one dose of the vac...