Recommended
While oral antivirals are not a substitute for vaccination, they help prevent severe illness, save lives, and preserve health systems. Vaccination rates continue to lag in low-income countries: only 14 percent of the population in low-income countries has received one dose of the vaccine, compared to 80 percent in high- and upper-middle-income countries. As uncertainties about future variants and vaccination campaigns persist, treatments must be elevated as a core strategy to combat COVID-19.
In a CGD paper launched today, we analyze the market landscape for two new oral antiviral therapeutics: a nirmatrelvir and ritonavir combination drug (Paxlovid), which was developed by Pfizer and reduces the relative risk of hospitalization by 89 percent, and molnupiravir, which was developed by Merck and reduces the relative risk of hospitalization by 30 percent. Both companies signed deals with the UN-backed Medicines Patent Pool (MPP) to allow some generic manufacturers to produce and sell the drugs in around 100 low-and-middle income countries. Companies selected by MPP for voluntary licensing with Pfizer and Merck were announced in March and January 2022 respectively. The sublicensees are geographically diverse, which enables greater supply chain adaptability, but the number of selected companies is also quite large (35 for nirmatrelvir/r and 27 for molnupiravir), which might lead to reduced capacity investments amid uncertain demand.
35 sublicenses signed with MPP for nirmatrelvir
Source: Medicines Patent Pool
27 sublicenses signed with MPP for molnupiravir
Source: Medicines Patent Pool
Given the range of bottlenecks, barriers to entry, demand inefficiencies, and gaps in testing infrastructure, there is considerable risk that production and uptake of oral antivirals will still be far too slow and modest in scale. Building from our analysis of the oral antiviral landscape, the paper offers near and medium-term actions the US government can take to ramp up manufacturing and accelerate access to promising COVID-19 treatments in LMICs. Here are a few of the main messages and recommendations.
1. We need better demand signals.
In the US context, a global oral antiviral access strategy—if adequately resourced—would first and foremost organize and generate demand. More certain demand will enable greater upfront investments in manufacturing capacity. Given that short-term access in LMICs depends on Pfizer and Merck’s production, the strategy should incorporate incentives for them to further scale-up manufacturing. To date, lawmakers have failed to deliver on a White House request for additional funding to fight COVID-19—including $5 billion for near-term international response efforts. If the US is to address gaps in therapeutic manufacturing incentives, the appropriation of new resources will be a crucial step.
Globally, the US government should create a consultative procurement forum with Africa CDC and the ACT-A therapeutics and diagnostics pillars, including the Global Fund, UNICEF Supply Division, UNITAD, FIND, WHO, and PAHO. This platform would help ensure that “market health” is given due importance in procurement awards by each of the relevant purchasers and facilitate information exchange and coordination on oral antiviral market dynamics. An existing external advisory group to the ACT-A therapeutics pillar management team could also be the right forum to discuss evolving demand forecasts.
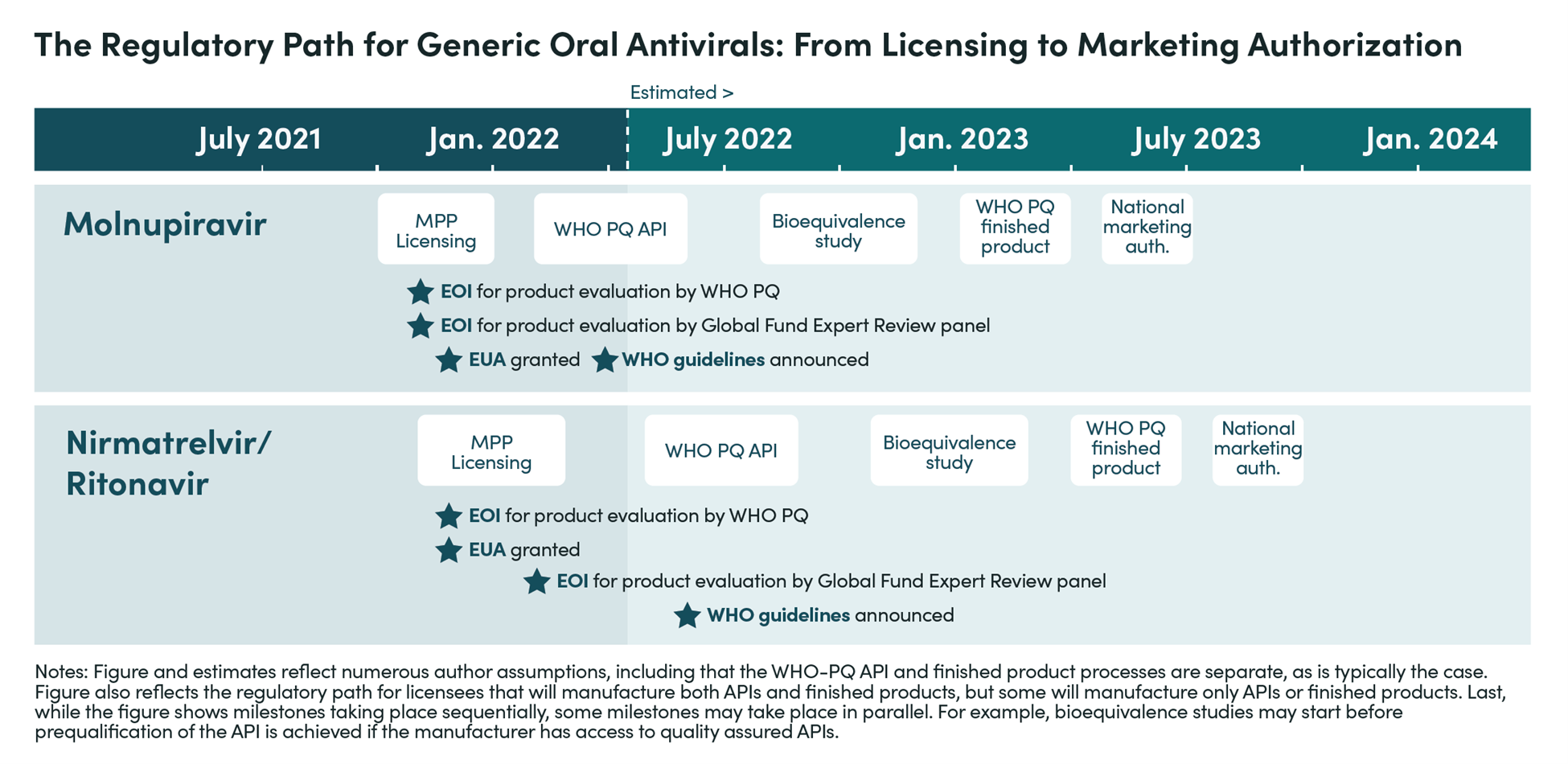
2. Based on interviews and analysis, we estimate that manufacturers for nirmatrelvir/r will begin filing for marketing authorization in LMICs—following approval—in 9 to 12 months under the best-case scenario, but will more realistically take up to 18 months unless sufficient policy actions are taken.
The short-to-medium term supply outlook thus depends crucially on availability from Pfizer. The below timeline notes the key steps that generic manufacturers must complete before filing for market authorization in LMICs.
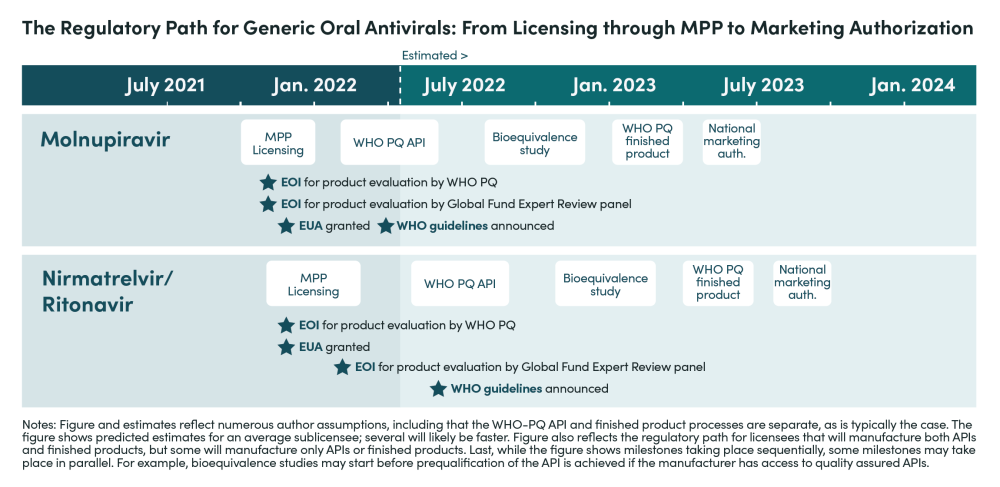
3. The US government can help reduce the time required for manufacturers to submit product dossiers for WHO prequalification (PQ) and reduce the length of the regulatory process itself.
First, USAID should leverage its implementing partner architecture and partnership with US Pharmacopeia to enhance technical support for generic manufacturers with voluntary licenses. As investments in process intensification and new formulation pathways yield results, technical assistance could help disseminate and spur more rapid use of such developments.
Second, the FDA should expand the conditional approval pathway currently used for HIV medicines to include generic oral antivirals for COVID-19 to be delivered outside of the US. This would increase global regulatory capacity to assess oral antivirals manufactured by companies with voluntary licensing agreements, strengthen the WHO PQ program, and reduce the time for products to reach LMIC markets. Additional support may be required for the FDA to dedicate resources towards this effort.
4. Existing foreign assistance architecture should be utilized for implementation.
Specifically, USAID can build on PEPFAR and PMI implementing partner infrastructure to create country deployment programs for test-treat initiatives, helping address testing and drug-drug interaction (DDI) management challenges. The US domestic test and treat strategy involves identifying ways to manage the complexities of DDIs for some of the oral antivirals. These efforts should be adapted quickly for use in resource limited health systems. PEPFAR and PMI implementers with significant experience supporting HIV, malaria, and TB test and treat in LMICs could then be resourced to initiate testing and treatment programs by providing countries the relevant tools and technical resources.
The US government can also partner with Pfizer and Merck to ensure needs-based allocation of limited supplies of oral antivirals using the country deployment plans. Such a partnership should define a single point of interface for the companies across different agencies working on COVID therapeutics.
Check out the full paper for more details, analytical insights, and priority actions. And stay tuned for more from CGD on therapeutics for COVID-19 and future outbreak countermeasures in the coming year.
DISCLAIMER & PERMISSIONS
CGD's publications reflect the views of the authors, drawing on prior research and experience in their areas of expertise. CGD is a nonpartisan, independent organization and does not take institutional positions. You may use and disseminate CGD's publications under these conditions.