Recommended
The US pays—by far—the highest prices for on-patent prescription drugs of any country on earth. The White House just issued an executive order purporting to fix this problem.
But the proposed solution—that the US should pay the “most-favored-nation” price for prescription drugs to “bring prices for American patients in line with comparably developed nations”—is misguided.
While that plan may sound reasonable at first hearing, it would do little to lower prices for US consumers. Instead, it would deny citizens of other countries access to lifesaving medicines they’d willingly pay for, and reduce incentives for research and development (R&D) of new drugs. The plan is motivated by a misunderstanding about why drug prices in the US are so high in the first place. (Notably, the order stops short of suggesting reference pricing against all countries, including poor ones, which is a good thing.)
Yes, the US pays too much for prescription drugs—and the pharmaceutical sector has long pushed back on price regulation
The US really does pay very high prices for on-patent prescription drugs—about four times as much as peer countries like Canada, the United Kingdom, France, and Japan. And as the order correctly points out, these high prices coexist with direct subsidy of upstream, publicly funded R&D—which helps pharma companies develop the same drugs they then sell back to the US market at high prices.
This executive order is the latest in a series of bipartisan efforts to lower drug prices. The first Trump administration tried to introduce caps on a subset of drugs covered by Medicare based on overseas prices (an approach blocked by the courts). The Biden administration then passed legislation, under the Inflation Reduction Act, allowing Medicare to negotiate lower prices for select drugs.
Meanwhile, the pharmaceutical industry has systematically fought efforts to negotiate or lower prices for Americans. That should come as no surprise: it is fundamentally a profit-maximizing industry with marketing and campaign contribution budgets in the billions. But the industry operates in a unique sector, and the executive order doesn’t account for that—there’s no single “fair” market price for pharmaceuticals.
Pharmaceutical companies are patent-protected monopolists, with profit-maximizing pricing strategies
Once drugs are developed, pharmaceutical companies want to pursue monopoly pricing strategies for their on-patent drugs. They also want to price discriminate to maximize overall profit. If they can only set a single price that applies worldwide, they will try to set a global profit-maximizing price. If they can set a different price in each market or country, they will try to set specific profit-maximizing prices for each country.
Meanwhile, drug buyers are usually governments with “monopsony power” (they’re by far the most significant purchaser in a given economy). Governments can decide on their maximum willingness to pay for a given drug, and demand access at that price. A pharmaceutical company is not obligated to sell at that price, but the alternative is to walk away and lose whatever the difference is between their marginal cost of production and the offered price.
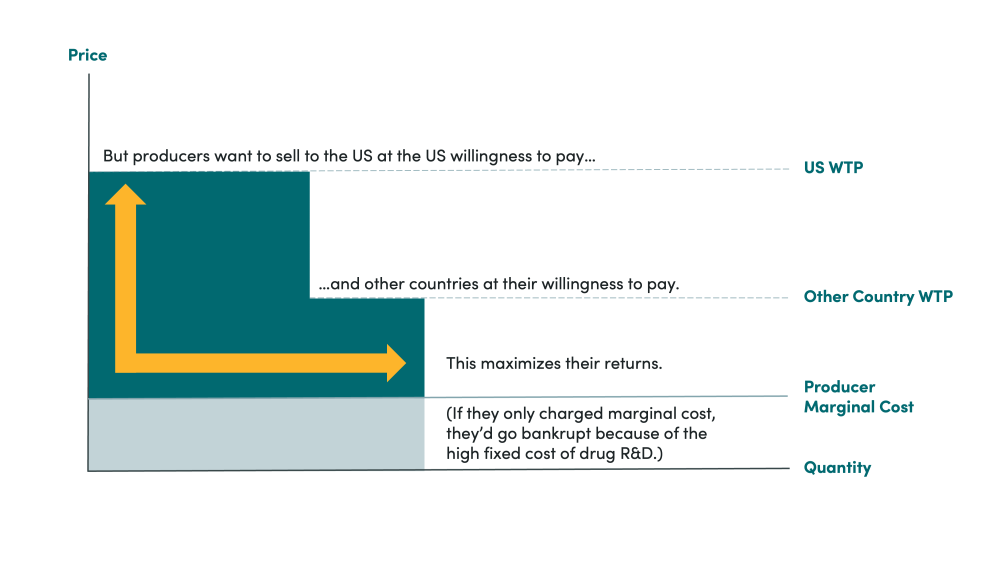
What does this mean in practice? Other countries are not receiving “discounts” against the US “real” price. Instead, everyone is paying prices somewhere below their willingness to pay (the value of the drug to the purchaser) and somewhere above the marginal cost of producing the drug (the minimum price it is worth selling at for the producer). Other countries are simply much less willing to pay the same amount for those drugs as US purchasers, so if they were offered the same price as the US, they frequently just wouldn’t buy the drugs (see figure 1).
What accounts for differences in willingness to pay across countries? Usually, rich-country buyers have more money to spend on drugs, so a higher willingness to pay for a drug of given efficacy. It makes sense that consumers in the United States would pay more, on average, than consumers, say in Greece, on that basis. There are also political differences—countries have different preferences and distinct purchasing policies and systems. America, for example, has a revealed willingness to pay for increased health that’s about 2-3 times higher than that of economically similar peers.
It is also important to note drug producers have considerable fixed costs related to R&D and regulatory approval but (usually) incredibly low marginal costs of actually producing pills. If every purchaser tries to push their price to the marginal cost of producing a pill, no company will develop new drugs, because they won’t be able to recoup those up-front costs. There is a tradeoff here: lower prices for available drugs means less incentive to develop new drugs. We’ve seen that in other parts of the health industry: when Medicare capped medical device prices, the result was a 25 percent decline in new product introductions and a 75 percent decrease in patent filings in the sector.
Figure 1. Comparison of willingness to pay for on-patent pharmaceuticals across countries

For the US, reference pricing makes little sense
“Reference pricing” is one way to negotiate prices—by refusing to pay more than the average or the minimum of some other set of countries. The White House proposal of “most favored nation” pricing, in line with “comparably developed nations,” is one such model. The executive order suggests that the US is carrying more than its fair share of the global fixed costs of new drug development, and reference pricing is a way to rebalance that.
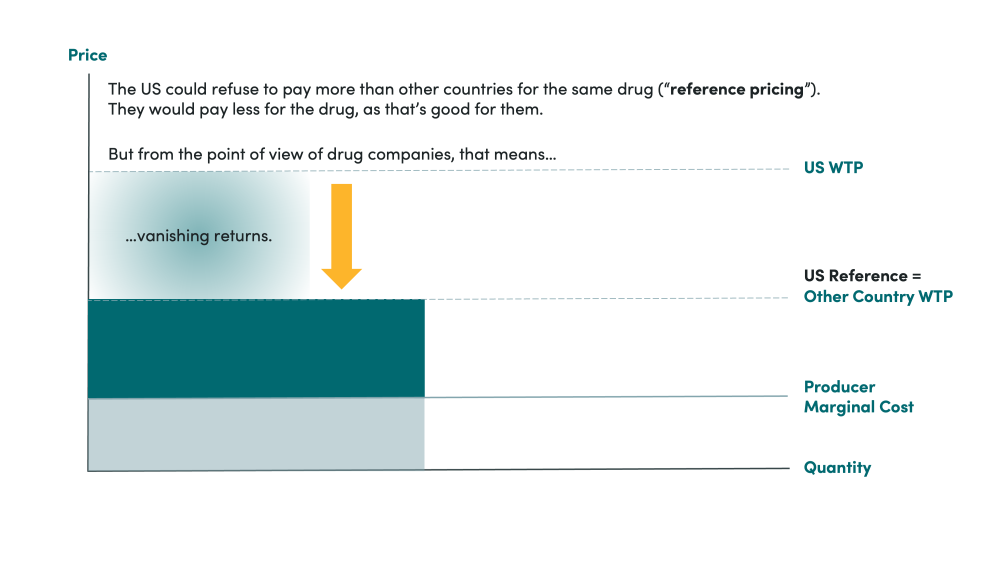
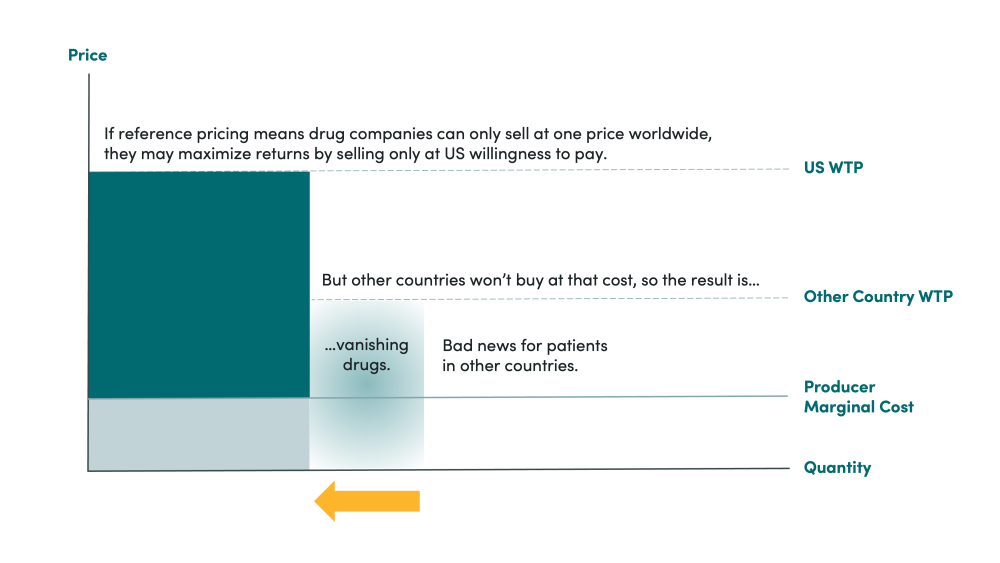
But the US market currently accounts for a huge share of pharmaceutical revenue and profits. Given the size of the US market, pharma companies will never willingly compromise their profits in this market to maintain sales in smaller, less important markets like Greece. So, if the administration demands, say, that the US pays the same price as Greece, pharmaceutical companies will likely simply raise the Greek price to the level America currently pays. America would likely still pay the same price, and Greece would walk away without access to medicines because it is not willing to pay the high American price. Essentially, pharma companies would no longer be able to price discriminate, and the US huge market share will shift the market to a single, global monopolist price.
This is a lose-lose-lose-lose (see figure 2): pharma companies will lose revenue and profits; Greeks (and people in many other countries around the world) will lose access to medicines; Americans will still be stuck paying high prices; and there will be less overall R&D incentive—meaning everyone worldwide loses access to a pipeline of new drugs.
Figure 2. Comparison of willingness to pay for on-patent pharmaceuticals across countries with reference pricing
Regulating prices should be about the US—not the rest of the world
Notwithstanding questions about how the administration would enact the proposed price reductions and whether it has the legal authority to do so, it is hard to say exactly what would happen under a US reference pricing regime. Referencing tends to be done using “ex-factory prices” provided by manufacturers, ignoring (sometimes significant) confidential discounts and rebates applied later. Small, poor countries may already pay more for drugs, and perhaps as a result the practical impact of reference pricing to date appears to be less than you might expect.
Nonetheless, the proposal to introduce “most favored nation” pricing incorrectly deflects blame for high American prices to other countries. Government purchasers in the US could use their monopsony power to rebalance prices paid in the US for drugs available today with incentives for new drugs tomorrow, but they should do that based on the efficacy and value that the drugs provide to US patients. (The administration might also want to look at ways to rein in pharmaceutical company marketing and patent evergreening strategies.)
The administration could also reasonably use trade negotiations to ask other countries to increase R&D spending on shared health priorities, but the efficient path to doing so is via direct, upstream public financing—not by pressuring them to pay higher prices that primarily benefit pharmaceutical companies. (It is estimated that just about a quarter of drug company revenue is reinvested into R&D, with the remainder going toward operating costs, marketing, lobbying, shareholder profit, and so forth.)
If enacted, the reference pricing policy would likely do little to nothing for drug prices here in the US. But it would deny patients access to drugs elsewhere, and reduce pharmaceutical company incentives to develop new drugs for patients in the US—and around the world.
DISCLAIMER & PERMISSIONS
CGD's publications reflect the views of the authors, drawing on prior research and experience in their areas of expertise. CGD is a nonpartisan, independent organization and does not take institutional positions. You may use and disseminate CGD's publications under these conditions.
Thumbnail image by: Chris Potter/Flickr






