Recommended
This brief is the second in CGD’s Tough Times, Tough Choices series, which offers guidance to major global health and development institutions and their funders on how to allocate resources strategically and effectively in an era of aid austerity.
Key recommendations
Gavi 6.0 should prioritize a dual mandate:
- Stabilize core vaccine budgets in the poorest countries
- Transform the financing model over time through a radically simplified “New Compact Envelope Financing” approach, with country-led prioritization, domestic financing of top priority vaccines, and a single Gavi cash envelope to cover additional vaccines and technical assistance
Gavi can generate cost savings of ~$2.5 billion without compromising its core mandate by:
- Cutting direct funding to non-Gavi-eligible middle-income countries, and offering alternative forms of high-leverage support
- Scaling back Secretariat expenses
- Securing vaccine price reductions
- Reducing funding for the African Vaccine Manufacturing Accelerator
- Decreasing overall country allocations with an incremental rollout by eligibility phase
Introduction
Following last month’s high-level pledging summit, Gavi has secured over $9 billion for its 2026-2030 strategic period (Gavi 6.0)—roughly $2.5 billion (21 percent) short of its proposed $11.9 billion budget. The replenishment outcome is disappointing given Gavi’s strong business case: immunization often ranks among the “best buys” in global health, and Gavi has been remarkably successful in fulfilling this mission to date, helping to vaccinate over a billion children.
Yet given the context, the outcome was also unsurprising. Earlier this year, the Trump administration announced it would stop future US contributions to Gavi—a decision it reaffirmed at last month’s summit, though US development budget negotiations are still ongoing between Congress and the administration. The UK, Gavi’s largest historical donor, recently slashed its development budget from 0.5 to 0.3 percent of gross national income (GNI), while other G7 countries have made similar aid cuts. In turn, these overall aid contractions are reflected in reduced national pledges to Gavi 6.0 (Figure 1). Gavi’s shortfall thus occurs amid a broader health financing crisis, including the dissolution of USAID, severe budget cuts at the World Health Organization and other UN agencies, and large-scale bilateral aid cuts. Many of the poorest countries—those eligible for Gavi support—are now facing severe disruptions to health financing, service delivery, and supply chains.
The scale of the crisis has also increased the urgency of calls for major global health reform. Building on the 2023 Lusaka Agenda, country governments, civil society, and donors alike appear open to increasingly ambitious proposals aimed at breaking down silos, strengthening national governments’ autonomy, and reducing aid dependency. Gavi’s leadership has already demonstrated an admirable openness to “radical transformation” through its Gavi “Leap” initiative, aligned with principles of country centricity, country sovereignty, limited mandates, and finite lifespans.
In this brief—the second in the Tough Times, Tough Choices series—we examine Gavi’s decision space for managing its budget shortfall. We propose a path forward that can yield savings while preserving Gavi’s highest-impact lines of effort and setting the foundation for future transformation. At this critical juncture, Gavi’s Board and leadership must stretch scarce resources to fulfill a challenging double mandate: (1) stabilizing immunization outcomes and fiscal solvency in Gavi-eligible countries facing severe health financing shocks; and (2) laying the groundwork for a durable transformation via a radically simplified “New Compact Envelope Financing” policy. We urge the Board to act upon these recommendations as it steers the organization through a hard-nosed strategic prioritization process in an era of budget austerity.
Figure 1. Gavi pledges by top historical donors in 5.0 (2021-2025) vs. 6.0 (2026-2030)
Where Gavi spends its money: Vaccines, and everything else
To identify potential cuts and opportunities for transformation, it is essential to first understand the structure of Gavi’s current spending. Gavi’s overall budget has grown consistently over time, but the composition of Gavi’s spending has changed (Figure 2).
Figure 2. Total budget by phase (2011-2030)
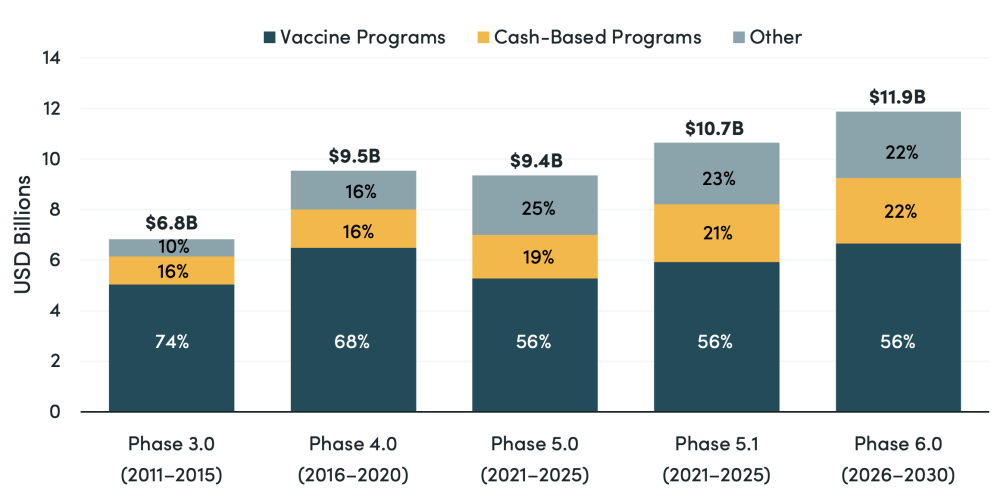
Notably, the budget for vaccine purchases has remained remarkably consistent since 2011, at around nominal $5 to 6 billion for each five-year period, with the majority allocated to high-impact, typically highly cost-effective vaccines. (After accounting for inflation, $5 billion in 2011 dollars is roughly equivalent to $7.3 billion today [i]—so in real terms, funding has fallen.) Gavi offers vaccine support to countries on a vaccine-by-vaccine basis; partner countries can select from Gavi’s “menu” of eligible vaccines and then pay a cofinancing fee for procured doses. This menu originally covered four diseases and has now expanded to 20, including the new malaria vaccines. The cofinancing requirement itself gradually increases as countries get wealthier and progress through the “initial self-financing,” “preparatory self-financing,” and “accelerated transition” phases, rising from a flat fee of $0.20 per dose in the poorest countries to nearly the full cost immediately prior to becoming “self-financing.” From a macro-budgetary perspective, over the past 15 years, inflation, new vaccine introductions, and population growth (cost drivers) have been roughly offset by vaccine price reductions, increased cofinancing revenue, and country transitions.
Beyond vaccine support, budgets for cash-grant support [ii] and other expenses have ballooned almost threefold, from $1.8 billion for Gavi 3.0 to a proposed $5.2 billion for Gavi 6.0. While not inherently negative, this evolution has led to an increased proliferation and complexity of funding windows, envelopes, and mechanisms. These include support for MICs, the African Vaccine Manufacturing Accelerator, the Partners’ Engagement Framework, and eight funding streams for cash-based programs supporting in-country immunization systems, such as health systems strengthening (HSS), vaccine introduction grants, and campaign operational costs (Table 1).
Table 1. Gavi offers funding to countries and partners under a range of different windows and mechanisms [non-exhaustive]
| Type | Description | Budgeted Amount for 6.0 (USD millions) |
|---|---|---|
| Cash-Grant Support | 2,604 | |
| Health systems strengthening | Includes support for countries to strengthen supply chains, build capacity for future vaccine introductions, and improve the equity, quality, efficiency, and sustainability of in-country immunization programs, and is intended to supplement domestic health system investments | 1,609 |
| Campaign operational costs | Covers campaign-related costs to facilitate the timely and effective delivery of vaccines to target populations | 863 |
| Vaccine introduction grants | Covers time-limited costs of a new vaccine to facilitate the timely and successful introduction into routine immunization programs | 118 |
| All other cash (ISS, INS, CSO, Switch Grant, etc.) | E.g., Switch Grants cover a share of one-off costs to switch product, presentation, schedule, or use | 14 |
| Other (select, non-comprehensive) | ||
| Catalytic support for MICs | Includes support for vaccine programs, technical and cash support to 1) prevent backsliding in vaccine coverage in former Gavi-eligible countries; and 2) drive introduction of key missing vaccines in both former and select never Gavi-eligible countries | 250 |
| Partners’ Engagement Framework (PEF) | Funding for a range of partners to support countries’ immunization programs; support is divided into three areas: 1) targeted country assistance (where most PEF funding is allocated); 2) special investments in strategic focus areas like supply chain, data, etc.; and 3) foundational support to partners such as WHO, UNICEF, World Bank, and others | 1,144 |
| ELTRACO (Eligibility, Transition & Co-financing) expenditure | Represents a mix of co-financing reductions and cash-grant support | 350 |
| African Vaccine Manufacturing Accelerator (AVMA) | A new financing instrument, approved in June 2024, which offers incentive payments to African manufacturers to promote the sustainable growth of Africa’s manufacturing base | 1,200* |
| Operating expenses | Consists of operating expenses for the Secretariat | 817 |
Notes: *AVMA is not reflected as a budget line item in Gavi 6.0; however, it represents earmarked funds over a 10-year period.
Sources: Gavi 6.0 Vaccine Investment Opportunity (Annex 3); Gavi’s MICs Approach; Gavi’s Partners’ Engagement Framework; Gavi’s African Vaccine Manufacturing Accelerator
Gavi’s vaccine support overwhelmingly goes toward highly cost-effective vaccines, but this can be a double-edged sword from a sustainability perspective. To date, Gavi’s cofinancing has operated on a vaccine-by-vaccine basis, providing a strong incentive to fulfill Gavi’s (gradually escalating) cofinancing requirements with domestic budgets (because these contributions are highly leveraged). Still, it also creates a strong disincentive for countries to support any individual vaccine exclusively with national funds (because this would imply losing a non-fungible Gavi subsidy)—meaning the entire vaccine program may remain dependent (to some extent) on donor financing.
The structural path forward: Stabilization and transformation
Earlier, we set out a two-pronged mandate for Gavi 6.0: (1) stabilizing distressed partner countries by protecting core immunization support, and (2) laying the groundwork for long-term transformation. Here, we outline the basic principles for operationalizing this mandate into practical prioritization decisions, before examining the budget math and identifying areas to cut in subsequent sections. We contend that a less strategic approach, particularly across-the-board cuts, could hinder Gavi’s impact over the next five years and undermine efforts to drive lasting transformation of global health institutions for long-term sustainability.
Stabilization
In the immediate term, Gavi must protect the poorest countries from further immediate financing or delivery disruption. Over the next two years—while countries absorb severe funding and supply chain disruptions due to cuts from other aid mechanisms—Gavi should ensure consistency and continuity for the poorest countries, e.g., the 43 countries in its portfolio (26 initial self-financing and 17 preparatory transition phase). These countries currently use Gavi support to finance the highest value and most cost-effective vaccines, including pneumococcal conjugate vaccine (PCV); inactivated polio vaccine (IPV); malaria vaccine; and measles, mumps, and rubella vaccine (MMR), as well as related campaign, health systems strengthening, and technical assistance costs to support vaccine delivery and immunization systems. These activities must continue uninterrupted in the immediate term to protect immunization outcomes and prevent avoidable, additional deaths; short-term continuity of process will also prove easier for country officials already struggling to react to severe USAID disruptions. However, this two-year stabilization period can be used to set the groundwork for future reforms, discussed below.
Transformation
First, Gavi should gradually adopt a flexible and radically simplified “new compact envelope financing” policy to structure its financing, with faster roll-out in transitioning countries. This policy should follow the principles of the “New Compact”—country-led prioritization, domestic financing of top priorities, and donor funding of the next priorities. At the highest level, this means that countries would move from the existing vaccine support plus cash grant support to a single, multi-year envelope allocation that could be flexibly used to support eligible vaccine procurement through Gavi/UNICEF and/or other immunization investments.[iii] Countries would have almost complete autonomy to use the allocation to support country-determined vaccine priorities, subject to a few conditions:
- To access the envelope funding, countries in the preparatory and accelerated transition groups must use their own resources to achieve (realistically aspirational) minimum coverage of a basic package of highly cost-effective vaccines (e.g., indicatively, 80 percent or higher coverage of PCV3, DTP3, and MMR, though the specifics should be determined following a context-specific, consultative, inclusive, and evidence-based process involving the Gavi Board, Secretariat, partner countries, and technical agencies). Countries in this group that domestically procure the basic vaccination package but fall short on coverage must use part of their envelope financing for interventions to increase effective coverage, subject to monitoring. To meet this condition, countries in the initial self-financing group should have progressive milestones that help incentivize progression over a five-year period; in the interim, countries may use their envelope financing to support the basic vaccine package, subject to pre-existing cofinancing requirements.
- The list of domestically financed vaccines would increase in size as countries become wealthier over the longer term, and the size of the envelope financing would decrease. This would be built into a revised Gavi’s Eligibility and Transition and Co-financing (ELTRACO) policy.
- Purchases of additional vaccines using the envelope funding must be procured through UNICEF, at least in the immediate term, to protect Gavi’s market shaping function. In the longer term, Gavi should consider increased optionality to procure through additional channels, for example the proposed Africa CDC Pooled Procurement Mechanism.
- Countries may use their allocation to procure vaccination-related technical assistance from a pre-approved list of providers.
- Countries may choose to access cash support for immunization systems by “pooling” it through a multilateral development bank (MDB) operation, appended as grant funding to broader health sector lending operations. This approach has multiple benefits: it helps bring grant financing on budget and promotes integration with national budget systems, while also leveraging MDB concessional resources for immunization, alongside other health priorities.
Second, Gavi should immediately move “accelerated transition” countries to “new compact envelope financing,” with other countries shifting to this after the stabilization phase. For current “accelerated transition” countries, immediate country envelopes could be smaller than what countries would receive under the current model, resulting in modest savings. Additional savings may then be achieved through annual reductions in their envelope allocation, potentially including a reduction in the transition timeline from eight to five years. The Secretariat should develop a comprehensive plan based on regular analysis of countries’ fiscal vulnerabilities, including other aid cuts and debt distress,[iv] and should develop a learning agenda from these “pilots” to inform broader roll-out of the new system in following years. Remaining “initial self-financing” and “preparatory transition” countries should begin moving to envelope financing after a two-year stabilization process, commencing in 2028, but they should not experience any immediate funding cuts compared to their status quo treatment.
The overall shift to “New Compact Envelope Financing” would have several benefits. It would:
- meaningfully operationalize a simpler, leaner, and more country-centric approach aligned with the Lusaka Agenda, allowing countries to align Gavi’s support more closely with domestic immunization priorities, and likely substantially reducing administrative burden and transaction costs for countries and the Secretariat, alike.
- provide a pathway (via an MDB) for putting cash support on budget, thereby funding in a way that directly supports country-owned health systems.
- incentivize governments to put the most basic, cost-effective vaccines on budget—and increase the strength of that incentive as they progress through transition phases—aligned with a “New Compact” approach.
- help counteract the effect of reduced overall funds as countries move through progressive stages of transition.
- facilitate more aggressive global health institutional reform in the years to come, e.g., potential consolidation of multiple competing financing mechanisms into a single health concessional financing fund.
Balancing the budget: Five areas for potential cost-cutting
We identify five areas below where Gavi can reduce spending from existing plans, amounting to roughly $2.5 billion, without severely compromising its core mandate—and while ensuring vaccine support to the poorest countries remains fully funded in the short term. We emphasize that while we view these as relatively low-impact and manageable cuts, they are generally not “costless” efficiencies—they are real strategic prioritization decisions that entail genuine trade-offs.
We also caution that our modelled estimates of savings are based on (limited) publicly available spending and budget data; our calculations may therefore differ from Gavi’s internal figures. We offer these suggestions (with indicative savings noted in parentheses) as a starting point for Secretariat and Board deliberations, to be supplemented by a more detailed exercise that pairs internal budget information with country fiscal analysis.
1. Cut direct funding to non-Gavi MICs and offer alternate forms of high-leverage support ($200 million)
Currently, Gavi offers a $250 million grant window to middle-income countries (MICs) that were previously eligible or never eligible for direct Gavi vaccine support. Although this support is intended to be catalytic, it remains relatively small-scale and ad hoc—and while it may be valuable in some cases, it cannot be justified in most MICs given the current funding crisis, since there are higher priorities in lower-income countries that are less able to fund their own vaccine programs. Gavi should consider eliminating most direct funding to these countries (at least for now) and instead providing high-leverage support with minimal budget impact. Modest exceptions should be made for emergency contexts of severe conflict and fragility, including countries hosting substantial refugee populations.
We suggest two alternate lines of effort that would benefit non-Gavi MICs: (1) market shaping and pricing support, including negotiation of opt-in framework agreements in which MICs can participate to procure vaccines (see this CGD paper), and (2) collaborating more systematically with MDBs to help countries unlock additional concessional resources for immunization. Doing so would require only extremely modest Secretariat resources.
Our proposal would sustain $50 million for countries in emergencies or experiencing instability as well as additional Secretariat activities, narrowing the budget shortfall by $200 million.[v]
2. Scale back Secretariat expenses ($280 million)
Operating costs were initially budgeted at over $800 million for Gavi 6.0. Gavi’s leadership has already committed to “radical simplification of the Secretariat,” to reduce costs by at least 30 percent by 2026 (yielding $240 million in savings across the entire five-year period). The Secretariat must ensure that cuts are strategic, preserving its core capabilities and ability to execute future reforms, as agreed by Secretariat leadership and the Board—and significant Secretariat cuts should not precede high-level Gavi-wide prioritization decisions. We recommend backloading these cuts to enable the Secretariat to manage the stabilization and transition period effectively.
Given recent growth in the Secretariat budget, we also note that a 30-percent cut essentially returns the Secretariat to pre-COVID capacity levels; it may thus be warranted to consider significantly deeper backloaded cuts, particularly given potential efficiencies from radically simplifying Secretariat processes, including the shift to an “envelope financing” approach (discussed below). We urge the Board to consider whether further cuts are possible without compromising core operations; for now, we budget based on a total cut of $280 million (35 percent)—slightly deeper than the 30-percent cuts already announced.
3. Secure vaccine price reductions ($200 million)
Already, Gavi has secured $200 million in anticipated cost savings for the 2026-2030 period through lower negotiated vaccine prices for malaria and rotavirus—an achievement announced at the pledging summit. As additional price reductions are not guaranteed, our budget math only reflects the $200 million in savings that have already been secured.
4. Reduce funding for the African Vaccine Manufacturing Accelerator ($900 million)
In June 2024, Gavi launched a $1.2 billion effort over 10 years, including leftover COVAX funds, to boost diversified manufacturing on the African continent—part of a politically salient broader effort to support African manufacturing following inequitable distribution of COVID-19 vaccines. The financing mechanism offers downstream incentive payments to manufacturers reaching specified “trigger points” to offset development costs, thereby “pulling” African-made vaccines to market. While the broader goal is both legitimate and important, we and others have previously noted serious conceptual and design challenges with the AVMA as currently proposed. Furthermore, partly due to its nascent stage, there is still no conclusive evidence on whether and to what extent it is contributing to its stated objectives.
Given the current funding shortfall, there is a strong argument to focus on Gavi’s core mission by delaying or eliminating some or all AVMA funding. This is, at core, a tough choice, potentially trading off immediate needs against long-term resilience. It may also be possible to achieve some savings without entirely compromising the program’s overall structure; considering realistic timelines for building commercially viable manufacturing capacity in Africa, it is possible that most earmarked resources won't be paid out until closer to the end of the 10-year program window, potentially freeing up some resources for allocation in the next five years.
We understand this initiative is politically sensitive; nevertheless, given that there is no existing evidence that the initiative will save lives cost-effectively, we encourage Gavi to seriously consider whether the AVMA is the best possible use of its scarce resources in the current climate. A relatively radical path forward would eliminate the initiative, yielding $1.2 billion in cost savings. A more measured approach—reflected in our headline calculations—would preserve $300 million for payments during Gavi 6.0, while reallocating the remaining $900 million for immediate use elsewhere.
5. Decrease overall country allocations with an incremental roll out by eligibility phase ($900 million)
In addition to the above measures, Gavi must also make additional cuts across various budget lines as it adopts a single “New Compact Envelope Financing” approach. Under our proposal, the different sources of money will eventually be consolidated into a single financing envelope, rendering the source of cuts irrelevant. However, specific pots of funds are disaggregated across distinct lines in the current proposed budget for Gavi 6.0 and are allocated across countries according to different criteria. Below, we provide an indicative set of proposed cuts across these existing line items to help balance the proposed 6.0 budget; we specify the source of cuts because this implies the level of total cut that countries at different stages in the transition continuum will experience.
ELTRACO ($350 million): Our proposed shift to an “New Compact Envelope Financing” approach would significantly change countries’ cofinancing requirements. Under this proposed approach, countries would not face explicit or “hard” cofinancing requirements as a portion of vaccine prices; they would merely need to ensure that the basic vaccine package is covered with domestic funds to access the remaining flexible envelope. In theory, this shift should obviate the need for explicit changes to the ELTRACO policy with budgetary implications, including cofinancing reductions and additional cash grant support; we therefore propose that the $350 million allocation for Gavi 6.0 [vi] can be eliminated. In practice, doing so will represent a significant cut to overall country allocations compared to the status quo, disproportionately affecting (relatively wealthier) countries in the accelerated transition group.
The Board should also revisit its December 2022 decision to extend the accelerated transition phase, which historically has lasted five years and was recently extended to eight. The shift to full New Compact envelope financing for accelerated transition countries, starting in 2026, may facilitate a faster five-year transition period, potentially with carve-outs for specific vaccines (e.g., malaria), which could yield additional savings for Gavi’s budget. For now, however, our calculations assume that the existing eight-year transition period will remain in place.
- Cash-grant support, including HSS ($400 million): The Gavi 6.0 budget contains a combined $2.6 billion in cash grant support, spread between HSS, vaccine introduction grants, campaign operational costs, and other much smaller categories. Alongside the shift to a single consolidated envelope approach, we propose fully funding the vaccine support budgets in all countries, while implementing a modest cut of $400 million (15 percent) to current allocations of cash-based support; the latter cuts should be backloaded for the 2028-2030 period and scaled by eligibility phase. The following provides an illustrative approach that could yield approximately $400 million in budget savings based on our modelling:
| Stabilization phase: 2026 – 2027 | Transformation phase: 2028-2030 | |
|---|---|---|
| Initial self-financing countries | No change above already-planned consolidation of cash-based grants into a single envelope | Move to envelope financing approach; envelope calculated as full vaccine support budget combined with 20 percent reduction in cash support |
| Preparatory transition countries | Move to envelope financing approach; envelope calculated as full vaccine support budget combined with 30 percent reduction in cash support | |
| Accelerated transition countries | Move to envelope financing approach; envelope calculated as full vaccine support budget combined with 20 percentreduction in cash support | Continue envelope financing approach; envelope calculated as full vaccine support budget combined with 35 percent reduction in cash-based support |
Partners’ Engagement Framework ($150 million): The Partners’ Engagement Framework (PEF) funds a range of partners, including WHO and UNICEF, as well as others like PATH, JSI, and the Clinton Health Access Initiative to a lesser extent, to support a mix of technical assistance and improvements in data and supply chain, among other areas, at the global and country levels. Some portion of PEF resources are allocated for targeted country assistance, with the rest spent at the global level; under Gavi 5.0, the country-specific ceilings for all countries for 2021-2025 totaled over $460 million (approximately half of the $913 million estimated for PEF).[vii] Under the envelope financing approach, the planned country-level PEF allocations for targeted country assistance should be bundled into the overall country financing envelopes; given reductions to country cash-based support proposed above, we argue against further reductions here.
Instead, Gavi should make a modest cut in global-level funding to partners, recovering $150 million from budgeted levels. Importantly, other partners, including the WHO and UNICEF, are facing their own budget crises, meaning sudden cuts could have additional negative ripple effects; however, this proposed cut represents just a $33 million (7 percent) reduction from Gavi 5.0 funding levels over five years, which should be possible to absorb without undue harm. [viii]
We recognize that these measures would entail reductions in country allocations. However, we expect countries will be able to find savings and efficiencies to reduce the harm of these cuts by
- flexibly allocating resources between vaccine support and other HSS and related objectives through the integration of the vaccine support and cash-based grant into a single envelope.
- flexibly allocating resources across different objectives (e.g., HSS and related immunization delivery areas) from the single cash envelope.
- “pooling” or “blending” (a portion of) envelope financing with MDB support to reduce external dependency for service delivery and longer-term systems improvements.
- identifying opportunities to procure technical assistance from more cost-effective providers outside of the Gavi partnership.
Figure 3. Summary of proposed path forward for Gavi 6.0
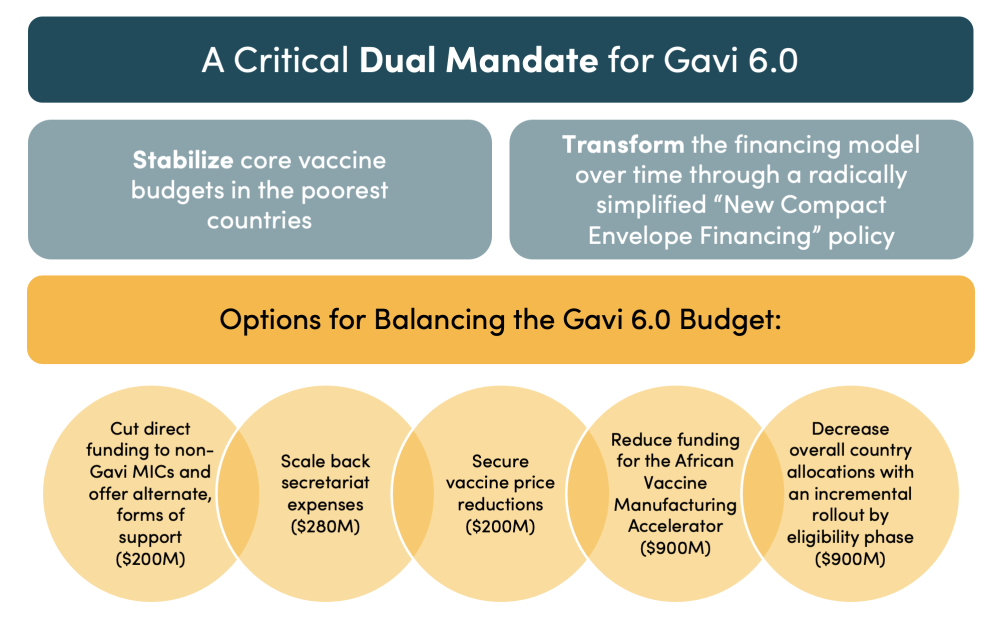
Conclusion
As Gavi operationalizes its next strategy, it must focus on achieving a critical dual mandate: (1) stabilizing and protecting the poorest countries from further immediate financing or delivery disruption; and (2) transforming its financing model via a radically simplified “New Compact Envelope Financing” approach aligned with core principles of the “Gavi Leap.”
We have proposed a viable and feasible path to do exactly that—all while saving the $2.5 billion needed for budget solvency. We urge the Board and Secretariat to align behind this clear vision for a bigger leap into Gavi’s future—one that meets the moment of our current crisis while also laying the foundation for real sustainability.
Thanks to Rosie Eldridge for research support, and to Adrian Gheorghe and Charles Kenny for feedback and suggestions on an earlier draft.
We invite interested individuals to propose a 300-word response article with clear counter proposals, which we plan to publish as a collection later in the year.
[i] Country ceilings for PEF for 2021-2025 are taken from here: https://www.gavi.org/sites/default/files/support/Gavi-5_0-Ceilings-by-country-and-support-type.pdf; Total PEF budget for Gavi 5.0 is here (Annex 4): https://reliefweb.int/report/world/prevent-protect-prosper-2021-2025-investment-opportunity
[ii] Global allocations for Gavi 5.0 totaled $453 million. The total PEF budget for 6.0 is $1.14 billion; assuming roughly half of that is allocated to in-country support, that implies a budgeted $570 million for global partners -- $117 million above prior the Gavi 5.0 level. A $150 million cut from budgeted levels would therefore represent just a $33 million cut from 5.0 enacted levels.
[iii] Eight countries currently eligible under Gavi’s MICs Approach are on the FY26 list of “Fragile and Conflicted-Affected” Situations, see here for countries eligible under Gavi’s MICs Approach and here for the FY26 FCAS list.
[iv] The $350 million represents an estimated cost to be incurred for Gavi 6.0 that results from decisions approved at the December 2024 board meeting to implement specific shifts to the ELTRACO policy, including reductions in cofinancing requirements. See here.
[v] This proposal is a more ambitious version of Gavi’s existing plans to combine eight separate non-vaccine cash support windows into a unified cash envelope for all countries, see https://www.gavi.org/sites/default/files/board/minutes/2024/6-7-june05%20-%20Annex%20B%20-%20Narrative%20for%20Gavi%206%200%20the%20Alliance%202026.pdf
[vi] According to prior CGD analysis, two accelerated transition countries—Kenya and Laos—may require additional monitoring, see here: https://www.cgdev.org/blog/26-countries-are-most-vulnerable-us-global-health-aid-cuts-can-other-funders-bridge-gap
[vii] Calculated via https://www.bls.gov/data/inflation_calculator.htm.
[viii] Vaccine support directly finances procurement of Gavi-approved vaccine doses. Cash-grant support includes a range of other cash-based support that helps to finance vaccination campaigns, vaccine introductions, and health system strengthening.
Topics
CITATION
Madan Keller, Janeen, Rachel Bonnifield, Tom Drake, Pete Baker, and Orin Levine. 2025. How Gavi 6.0 Can Take a Bigger Leap. Center for Global Development.DISCLAIMER & PERMISSIONS
CGD's publications reflect the views of the authors, drawing on prior research and experience in their areas of expertise. CGD is a nonpartisan, independent organization and does not take institutional positions. You may use and disseminate CGD's publications under these conditions.